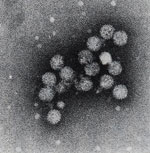
Diagnostics for the early detection and management of hepatitis C infection
Early detection of HCV infection is vital in order to provide consolidated care of acute and chronic infection, and adequate treatment of drug response monitoring is crucial to overall therapy success. Patients undergoing a peginterferon alfa-2a and ribavirin therapy can be monitored precisely by real-time PCR measurement. The novel Elecsys anti-HCV II immunoassay on cobas e modules allows the early qualitative detection of antibodies against HCV. The updated assay delivers enhanced convenience, specificity, increased reliability and consistency for improved efficiency in screening patients as well as blood donors. Following the identification of HCV antibodies, testing for HCV RNA, which is a crucial marker in the management of hepatitis C, confirms the infection and facilitates prediction of treatment response. Sensitive HCV RNA qualitative and quantitative tests to measure viral load and predict drug response are available on the COBAS AmpliPrep/COBAS TaqMan fully automated platform.
Supplier: Roche Diagnostics International Ltd.
Website: