Anti-laminin β4 – a new autoantibody marker in pemphigoid disease
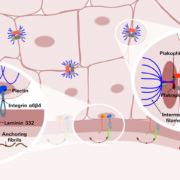
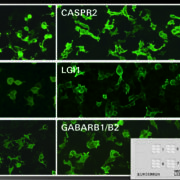
Laminin β4 (lamβ4) has been identified as a novel target antigen in anti-p200 pemphigoid, a rare autoimmune disease manifesting with subepidermal blisters. Anti-lamβ4 antibodies bind to the region of the dermal–epidermal junction and demonstrate pathogenic effects. A recombinant-cell indirect immunofluorescence assay has been developed for specific detection of anti-lamβ4 antibodies.