H. cinaedi: infection, detection and diagnosis
Helicobacter cinaedi is a relatively recently identified bacterium, but it is recognized as an increasingly important cause of disease in humans. This article summarizes methods for its detection and identification as well as routes of infection and treatment.
by Prof. Yoshiaki Kawamura, Dr Tatsuya Okamoto, Dr Shigemoto Fujii and Prof. Takaaki Akaike
What is Helicobacter cinaedi?
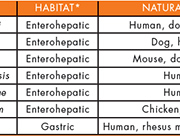
Within the genus Helicobacter, 33 species to date have been proposed and validated, but only 7 species have been isolated from human clinical specimens (Table 1). H. pylori, classified as a ‘gastric Helicobacter species’, is a well-known member of Helicobacter, but some less well-known ‘enterohepatic Helicobacter species’, such as H. cinaedi, H. bilis, H. canadensis, H. canis, H. fennelliae, and H. pullorum, have also been isolated from human clinical specimens.
H. cinaedi was first isolated as a Campylobacter-like organism type 1 (CLO-1) in 1984 from rectal swabs from homosexual men displaying intestinal symptoms [1] and the following year the organism was named ‘Campylobacter cinaedi’; however, it was subsequently reclassified as Helicobacter [2]. During the last two decades, there have been many reports of the isolation of H. cinaedi from blood or intestinal tract of human immunodeficiency virus-infected or immunocompromised patients, but, recently, increasing numbers of infections have also been reported in immunocompetent patients.
In Japan, the isolation of H. cinaedi was first reported in 2003; since then, its isolation has been reported from patients regardless of gender and within a wide age range, from the newborn to the elderly, in many hospitals throughout the country. We have experienced many cases of H. cinaedi cellulitis and bacteremia in both immunocompromised and immunocompetent subjects in hospitals. This microorganism should, therefore, be considered a causative agent of nosocomial infection [3].
Illnesses caused by H. cinaedi
Clinical symptoms of H. cinaedi infection include fever, diarrhoea, abdominal pain, gastroenteritis, proctitis, cellulitis, erysipelas, arthritis, meningitis, and bacteremia. In contrast to other Helicobacter species, numerous reports have causally linked H. cinaedi infection with bacteremia, which contributes to this organism’s strong vascular invasion ability. In many cases of H. cinaedi bacteremia, the main symptom is fever accompanied by arthritis and cellulitis at various sites. In addition to these sites providing a source of primary infection, the resultant bacteremia can serve as a source of secondary infections; thus, all these various symptoms are clinically important.
In our experience, at various times after orthopedic surgery (range, 8–113 days; mean, 29 days), some patients had a sudden onset of local flat cellulitis (salmon-pink in colour) at different sites on the operated side, along with fever and an increase in C-reactive protein level (Fig. 1) [3]. Cellulitis was often multifocal without wound infection. Many of these patients had been treated for fracture and were immunocompetent.
In recent years, we have demonstrated the potential association of H. cinaedi with atrial arrhythmias and atherosclerosis [4]. This could be through bacterial translocation of H. cinaedi from the intestinal tract into the blood stream. The possible cause-and-effect relationship between H. cinaedi and vascular diseases may warrant further epidemiological study on proatherosclerotic effect of H. cinaedi infection.
The virulence factor of H. cinaedi is largely unknown. The complete genome sequence of a human clinical isolate was announced in 2012 [5] and revealed that the organism holds a Type VI secretion system, which is expected to be related to its virulence, together with two known virulence factors, cytolethal distending toxin and alkyl hydroperoxide reductase.
Detection, cultivation and identification of H. cinaedi
It is well known that H. cinaedi is a fastidious and slow-growing organism, and that detection and cultivation are extremely difficult. In many cases, H. cinaedi is first detected from a blood culture using an automatic blood culture system. It is generally noted that 4–10 days (average 5.6 days, in our experience) are needed for a positive result in the culture bottle of an automatic blood culture system, such as the BACTEC system (Becton Dickinson) using an aerobic bottle. Therefore, when the culture test using this system is terminated within 3–4 days, the bacterial growth may be still below its detection limit. Information on the detection of H. cinaedi using the BacT/ALERT system (Biomérieux) is scanty. In our experience, the VersaTREK system (Thermo Scientific) is superior for the detection of this microorganism.
Both H. cinaedi and H. pylori are members of the genus Helicobacter; however, the former is extremely difficult to culture. H. cinaedi isolates essentially require microaerobic conditions (5–10% O2) and a high level of humidity. Often a blood agar plate stored in a refrigerator for a few days may fail to support the growth of H. cinaedi because of low water content. Use of fresh medium is strongly recommended. It is established that H. cinaedi growth is accelerated by adding hydrogen gas (5–10%) to microaerobic conditions. The culture success rate can be improved by using a gas mixture such as 6% O2, 7% H2, 7% CO2, and 80% N2 at the initial culture from the clinical specimen or in the culture bottle. Unfortunately, many of the commercially available microaerobic gas-generating packs, such as the GasPak system (Becton Dickinson), deoxidize and generate CO2 but do not generate hydrogen gas; therefore, in some cases H. cinaedi does not grow, or growth is inadequate.
H. cinaedi cultured on an agar plate grows in a film, which is difficult to identify visually. Therefore, the culture should be carefully checked on the plate.
The biochemical identification of this organism is problematic owing to unstable phenotypic reactions. In many cases commercially available identification kits do not produce reliable results. Therefore, identification based on nucleotide sequence or species-specific polymerase chain reaction (PCR) has been used. We have developed a nested PCR system with high specificity and sensitivity (approximately 102 CFU/ml) for detecting H. cinaedi [6]. We have also established an immunological diagnosis method (antibody detecting test) with high specificity to detect the exposure history of H. cinaedi [7].
Antimicrobial therapy and prognosis
To date, antimicrobial susceptibility testing for H. cinaedi has mainly used the agar-dilution method, but this method is too cumbersome for routine use in hospital laboratories. A broth microdilution method for antimicrobial susceptibility testing of H. cinaedi, which can be performed easily, has been developed by our research group [8].
H. cinaedi strains generally show low minimum inhibitory concentration (MIC) values for carbapenems, aminoglycosides, and tetracycline (MIC90 = 1 µg/ml for imipenem/cilastatin, gentamicin, and tetracycline). In contrast, H. cinaedi has well-known resistance to macrolides, with especially high MIC values (MIC90 >64 µg/ml for erythromycin). Recently in Japan and elsewhere, H. cinaedi isolates have shown high resistance to quinolones (MIC90 = 64 µg/ml for ciprofloxacin and levofloxacin) due to point mutation(s) of DNA gyrase genes [8].
Symptoms caused by H. cinaedi, such as fever or cellulitis, usually resolve after 2 to 3 days of drug therapy, but the Centers for Disease Control and Prevention recommended long-term therapy for about 2 to 6 weeks, rather than short-term therapy for only 10 days [9]. Prognosis is generally good, but it is noteworthy that, depending on the study, about 30–60% of patients have recurrent symptoms. Unfortunately, there are no guidelines for the treatment of H. cinaedi infections, including the clinical breakpoints of antimicrobial agents. The MIC values described above are based on our data.
Infection route
H. cinaedi has been found in a wide range of animals including cats, dogs, hamsters, rats, and foxes. There have been many reports of zoonotic transmission vectors, but no reports of the simultaneous isolation of H. cinaedi from human patients and the animals that they have been in close contact with. It is noteworthy that H. cinaedi isolates from human, dog, and hamster formed a distinct ribotype pattern group by host source [10].
Epidemiological analysis methods, such as pulse-field gel electrophoresis, randomly amplified polymorphic DNA, and multilocus sequence typing, have been proposed for H. cinaedi isolates [3, 11]. As described above, we developed a nested PCR system and immunological diagnosis method. Using these methods, we tested many healthy hospital employees (doctors, nurses, staff members, etc.) and found that some currently uninfected individuals had previously had H. cinaedi infections, indicating that there could be asymptomatic carriers with intestinal colonization of H. cinaedi. Our study also suggested that occurrence of such asymptomatic carriers may be related to nosocomial infection.
However, the complete route of infection route, including nosocomial transmission, of H. cinaedi remains unclear.
Summary
A full understanding of H. cinaedi infection remains elusive; however, some features and the clinical relevance of this infection have become increasingly recognized recently. To detect and isolate H. cinaedi from human blood samples using an automated blood culture system, a long-term incubation (up to 10 days) is needed and further skillful culture techniques are required. In many clinical laboratories, however, appropriate culture for isolation of this bacteria might not performed, which may lead to false-negative findings for H. cinaedi. As H. cinaedi was considered not to cause acute severe disease, it seems that its importance may have not been recognized clinically. However, we now know that this microorganism likely causes nosocomial infections that are difficult to eradicate and have a high incidence of recurrence. In recent years, a possible association with chronic illnesses such as arrhythmia and arteriosclerosis has been reported, and therefore we will need to carefully monitor and ascertain trends in H. cinaedi infections.
References
1. Fennell CL, et al. E. Characterization of Campylobacter-like organisms isolated from homosexual men. J Infect Dis. 1984; 149: 58–66.
2. Vandamme P, et al. Revision of Campylobacter, Helicobacter, and Wolinella taxonomy: emendation of generic descriptions and proposal of Arcobacter gen. nov. Int J Syst Bacteriol. 1991; 41: 88–103.
3. Kitamura T, et al. Helicobacter cinaedi cellulitis and bacteremia in immunocompetent hosts after orthopedic surgery. J Clin Microbiol. 2007; 45: 31–38.
4. Khan S, et al. Promotion of atherosclerosis by Helicobacter cinaedi infection that involves macrophage-driven proinflammatory responses. Sci Reports 2014; In press.
5. Goto T, et al. Complete genome sequence of Helicobacter cinaedi strain PAGU611, isolated in a case of human bacteremia. J Bacteriol. 2012; 194: 3744–3745.
6. Oyama K, et al. Identification of and screening for human Helicobacter cinaedi infections and carriers via nested PCR. J Clin Microbiol. 2012; 50: 3893–3900.
7. Iwashita H, et al. Identification of the major antigenic protein of Helicobacter cinaedi and its immunogenicity in humans with H. cinaedi infections. Clin Vaccine Immunol. 2008; 15: 513–521.
8. Tomida J, et al. Comparative evaluation of agar dilution and broth microdilution methods for antibiotic susceptibility testing of Helicobacter cinaedi. Microbiol Immunol. 2013; 57: 353–358.
9. Kiehlbauch JA, et al. Helicobacter cinaedi-associated bacteremia and cellulitis in immunocompromised patients. Ann Intern Med. 1994; 121: 90–93.
10. Kiehlbauch JA, et al. Genotypic and phenotypic characterization of Helicobacter cinaedi and Helicobacter fennelliae strains isolated from humans and animals. J Clin Microbiol. 1995; 33: 2940–2947.
11. Rimbara E, et al. Molecular epidemiologic analysis and antimicrobial resistance of Helicobacter cinaedi isolated from seven hospitals in Japan. J Clin Microbiol. 2012; 50: 2553–2560.
12. Solnick JV, Schauer DB. Emergence of diverse Helicobacter species in the pathogenesis of gastric and enterohepatic diseases Clin Microbiol Rev. 2001; 14: 59–97.
The authors
Yoshiaki Kawamura1 PhD; Tatsuya Okamoto2 MD, PhD; Shigemoto Fujii3 PhD; Takaaki Akaike3* MD, PhD
1Department of Microbiology, School of Pharmacy, Aichi Gakuin University, Nagoya, Japan
2Intensive Care Unit, National Center for Global Health and Medicine, Tokyo, Japan
3Department of Environmental Health Sciences and Molecular Toxicology, Tohoku University Graduate School of Medicine, Sendai, Japan
*Corresponding author
E-mail: takaike@med.tohoku.ac.jp