Bruker expands MALDI Biotyper and IR Biotyper platforms for microbial identification and outbreak management
Bruker has announced a series of portfolio enhancements to its MALDI Biotyper® (MBT) and IR Biotyper® systems, unveiled at ESCMID Global 2026 in Munich. The updates span microbial identification, automated sample preparation, outbreak surveillance, antimicrobial susceptibility testing (AST), and reflex next-generation sequencing (NGS) workflows – with both research-use-only (RUO) and IVDR-registered developments included.
Expanded reference libraries and a new consumables kit
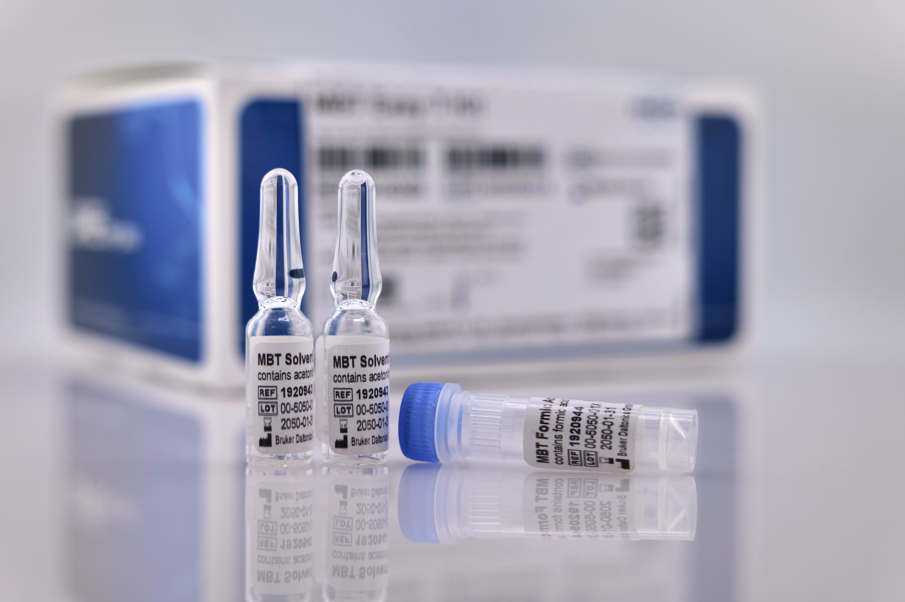
A central update for routine MALDI-TOF workflows is the introduction of the MBT Easy T® Kit (available as RUO and IVDR), a consumables kit designed to standardise sample transfer to MALDI target plates. The kit includes pre-filled, ready-to-use reagents and applicators – including formic acid ampoules – supporting up to 2,000 samples per kit, with room-temperature storage and reduced chemical waste.
Kirsten Schönfeld, Diplom-Biologin at Institut Dr. Nowak GmbH, Germany, commented: “The MBT Easy T Kit provides pre-filled, ready-to-use solvent ampoules, including formic acid, which will simplify logistics and daily laboratory operations. This helps maintain a robust and streamlined MALDI Biotyper workflow in a clinical setting.”
Bruker has also expanded its MALDI Biotyper reference libraries to more than 5,300 species, incorporating bacteria, yeasts, filamentous fungi, and mycobacteria (RUO). The update adds over 600 new species and includes approximately 30% stronger species coverage for the dedicated filamentous fungi MyT workflow. Corresponding IVDR-registered library expansions are described as currently in progress.
For specialist identification beyond local installations, Bruker is offering early key opinion leader access to a cloud-based MALDI identification solution (RUO), operating within the MBT Compass HT environment. Initial libraries include yeast collections, anaerobic bacteria libraries developed by academic partners, and mosquito libraries for malaria vector monitoring research. The cloud infrastructure also provides a basis for online system monitoring and software updates.
Automation and clinical validation studies
Two automated sample preparation systems – MBT PrepMatic™ for colony-based cultures and MBT SepsiMatic® for positive blood culture bottles – are currently undergoing IVDR-compliant clinical validation studies. MBT PrepMatic is designed to enable semi- to fully automated colony picking from Petri dish cultures, whilst MBT SepsiMatic supports a fully automated workflow from positive blood culture bottles through to prepared MALDI target plates, with the potential to yield purified samples containing viable cells for downstream analysis.
In the United States, clinical studies are under way to support upcoming MALDI Biotyper identification claims for mycobacteria and filamentous fungi, with FDA submissions planned for 2026.
Rapid AST and AI-based resistance prediction
Bruker is progressing clinical validation of MBT FAST, a rapid phenotypic AST approach using MALDI-TOF MS that aims to enable same-day results from positive blood cultures and agar colonies for Enterobacteriaceae. The methodology employs machine-learning-based growth assessment and aligns conceptually with CLSI- and EUCAST-guided broth microdilution principles.
IR Biotyper outbreak surveillance updates
The IR Biotyper platform (CE-marked, general purpose) has been expanded with IR Tracker for hospital-acquired infection (HAI) surveillance, alongside new classifiers for Salmonella Typhi, Shiga toxin-producing E. coli (STEC), and Shigella species – organisms associated with specific treatment requirements, high infectivity, and public health reporting obligations.
NGS integration and culture-independent diagnostics
A new software version of the MBioSEQ™ Ridom Typer (RUO) adds TB-Profiler-based antimicrobial resistance determination and GAMBIT bacterial species identification, supporting MALDI/IR Biotyper-triggered WGS reflex workflows. Separately, Bruker has integrated Molzym – acquired in late 2025 – into its Microbiology & Infection Diagnostics portfolio. Molzym’s MolYsis™ host-DNA depletion technology selectively removes host DNA to facilitate sensitive detection of microbial DNA in low-biomass and blood samples for downstream sequencing.
Dr Wolfgang Pusch, President of Bruker’s Microbiology & Infection Diagnostics Division, commented: “Bruker is pursuing a broad and sustained innovation initiative – significantly expanding our reference libraries, introducing new sample preparation options to enable the highest level of standardization, and advancing exploratory artificial-intelligence-based approaches for antibiotic resistance prediction from MALDI spectra.”
For more information, visit: www.bruker.com
Digital issue: Please click here for more information