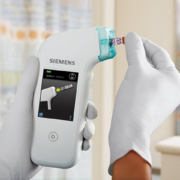
The Hemo Control point-of-care (POC) diagnostics analyser provides laboratory accurate hemoglobin and hematocrit results in one simple test. The new hemoglobin POC analyser enables full upgrade with data management functionality as required, giving the user complete flexibility over their future connectivity options via its bi-directional interface. Using the new LIS2-A2 communication standard, this bidirectional interface allows the analyser to connect directly to third party software and enable simple integration with LIS (Laboratory Information System) middleware. LIS2-A2 succeeds the ASTM communication standard commonly used for small to mid-size analysers and is easier to integrate than POCT-1A. Available as a cost effective basic device which stores up to 4000 patient results, the new Hemo Control offers enhanced on-board Data Management (DM) functions following upgrade with the DM ‘add pack’ if required at a later date. In addition to enabling lists to be sent directly from the LIS to Hemo Control, the DM upgrade also offers barcode identification of patients, operators, cuvette LOT and control materials, as well as quality control with lock-out functions and the ability to add comments to results. Hemo Control requires no docking station when handling on-board data management as it uses simple cable connections (e.g. RS-232 to USB or LAN), or integrated Bluetooth technology which is available with the Hemo Control Manager – the top of the range device available with preinstalled DM functions. As a highly robust, portable analyser, Hemo Control is designed to provide quantitative, lab quality results for both hemoglobin and hematocrit (imprecision of <2%) from 25 seconds for blood banks, hospitals and clinics. User friendly with step-by-step on-screen instructions and no calibration or maintenance needed, the device requires minimal training. Furthermore, this compact analyser uses NXT shape microcuvette technology, which enables improved finger stick sample collection by virtually eliminating appearance of air bubbles.
Read more