Complete laboratory diagnosis of Zika virus infections
Laboratory diagnosis of Zika virus (ZIKV) infections is based on two main pillars: direct detection of the viral RNA genome and serological detection of anti-ZIKV antibodies. Direct detection of the virus by reverse transcriptase real-time polymerase chain reaction (RT real-time PCR) is the most important method for diagnosing early acute infections. A new RT real-time PCR system with fully automated data evaluation provides highly standardized and streamlined detection of ZIKV RNA. Serology is useful for acute diagnostics as well as for longer term monitoring and epidemiological studies. An ELISA based on ZIKV NS1 antigen provides exceptionally high specificity with virtually no cross reactivity to other flaviviruses.
by Dr Jacqueline Gosink
Introduction
ZIKV has become firmly established in South and Central America and the Caribbean and is increasingly spreading to other parts of the world. The infection is now classified by the World Health Organization as an enduring public health challenge. Nearly one million people in 48 countries have been infected with ZIKV since the beginning of 2015, according to the Panamerican Health Organization. The actual number of cases is presumably much higher, since many infections are mild and go unreported. The virus is transmitted predominantly by mosquitos of the Aedes genus, which are ubiquitous in many topical and non-tropical regions. Transmission by sexual contact is also increasingly described. ZIKV infections are difficult to distinguish clinically from dengue virus (DENV) and chikungunya virus (CHIKV) infections, which manifest with similar symptoms of fever, exanthema and arthritis and are endemic in much the same geographic regions. There is, however, a growing body of evidence linking ZIKV to birth defects in fetuses and newborns and neurological complications such as Guillain-Barré syndrome in adults. Therefore, accurate diagnosis of ZIKV infections and differentiation between acute and past infections is critical for effective patient care.
ZIKV direct detection
The ZIKV RNA genome can be detected during the viremic phase of infection. The viral RNA is detectable for up to around 5 days after the onset of symptoms in serum and up to 10 days in urine. Molecular diagnostic detection is therefore highly effective for early diagnosis of ZIKV infections and discrimination of ZIKV from clinically similar infections such as DENV or CHIKV.
Novel RT real-time PCR assay
A new assay provides fast detection of ZIKV RNA in serum or urine by reverse transcriptase real-time polymerase chain reaction (RT real-time PCR) with fully automated data analysis. The EURORealTime Zika virus test is based on a one-tube reaction, comprising reverse transcription of the viral RNA into complementary DNA (cDNA) followed by PCR amplification and fluorescence-based real-time detection of defined sections of the ZIKV genome. The reverse transcription, amplification and detection of ZIKV cDNA are carried out by means of ZIKV-specific DNA primers and real-time DNA probes. RNA-based internal and positive controls verify the correct performance, integrity and functionality of the complete procedure. Ready-to-use reagents provide added reliability and convenience.
The evaluation of results is fully automated using the EURORealTime Analysis software and is therefore highly standardized and objective. All results, including those of the controls, are documented and archived. The software also supports simple and error-free test performance by guiding every step of the workflow. The entire detection procedure (excluding RNA extraction) takes less than 90 min.
Specifications and evaluation of the EURORealTime Zika virus test
Highest test sensitivity and specificity is ensured by the meticulous design of the primers and probes. Moreover, cross reactivity with other pathogens that may be present in serum or urine samples and/or are closely related to ZIKV has been excluded experimentally.
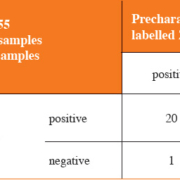
In clinical evaluation, 29 serum and 26 urine samples from patients with suspected ZIKV infection were analysed using the EURORealTime Zika virus and another CE/IVD-labelled ZIKV test system. There was a positive agreement of 95.2% and a negative agreement of 97.0% between the results obtained with the two tests (Table 1).
ZIKV serology
Serological detection is effective from soon after symptom onset (4-7 days) to beyond convalescence. Serology serves as a supplement to RT-PCR in acute cases. It is especially useful in cases where viral RNA is no longer detectable, for example if the infection is resolved or has moved into the chronic phase. Serological detection is particularly relevant in prenatal diagnostics, sexual healthcare and epidemiological surveys. Pregnant women with serological evidence of an infection can be offered intense prenatal monitoring, while seronegative women may be spared unnecessary worry. Due to the lengthy presence of ZIKV in semen, men who have resided in or travelled in endemic regions are advised to abstain from unprotected sexual intercourse for six months after returning to prevent sexual transmission, especially when their partner is or could be pregnant. Serological testing can be helpful in these cases for excluding or identifying an infection. As ZIKV continues to move into previously unaffected areas, epidemiological studies using serological methods can help to monitor the spread of the virus and probe its associated complications.
Relevance of immunoglobulin classes
Primary acute ZIKV infections are generally characterized by the occurrence of specific IgM antibodies, with IgG appearing at the same time or shortly afterwards. IgM can remain detectable for several months, while IgG is assumed to persist lifelong. Detection of specific IgM or a rise in the specific IgG titre in a pair of samples taken at least 7 to 10 days apart is evidence of an acute infection.
In secondary flavivirus infections, for example following a previous vaccination or infection with another flavivirus, specific IgM is often found at a low or undetectable titre. Therefore, additional tests like the detection of IgG or plaque reduction neutralization test are recommended.
Specific IgA may also be useful for diagnostics. In secondary flavivirus infections synthesis of IgG is rapidly stimulated. Shortly after infection the IgG titre levels off and is indistinguishable from IgG titres in convalescent infections, making seroconversion difficult to detect. This pattern has been observed in ZIKV patients from regions endemic for other flaviviruses. IgA has recently been proposed as a putative additional marker of acute infection in cases where IgM is not detectable and the IgG titre is already high.
Highly specific NS1-based ZIKV ELISA
Serological diagnosis of ZIKV is challenging due to the high cross-reactivity between flavivirus antibodies. This obstacle has been overcome by the use of recombinant non-structural protein 1 (NS1) from ZIKV as the antigenic substrate in ELISA. Use of this antigen avoids the cross-reactivity typically associated with tests based on whole virus antigens or viral glycoproteins. The NS1-based ELISA provides highly sensitive and specific ZIKV diagnostics, as demonstrated in numerous studies.
Clinical evaluation of IgM/IgG ELISA
The NS1-based Anti-Zika Virus ELISA was used to examine anti-ZIKV antibodies of classes IgG and IgM in various serum panels. In samples from patients with RT-PCR-confirmed infections (n=71), taken 5 days or more after symptom onset, the sensitivity of the test amounted to 100% for IgG/IgM (Table 2) (1). In a panel of blood donors the specificity of the ELISA was 99.8%.
In studies with a total of over 450 patients harbouring other arboviral infections, including DENV, CHIKV, tick-borne encephalitis virus (TBEV), West Nile virus (WNV), Japanese encephalitis virus (JEV), and individuals vaccinated against yellow fever virus (YFV) or TBEV, the specificity lay between 96% and 100% (Table 3) (1, 2). In particular, a specificity of 100% was observed in DENV- and CHIKV-infected patients, demonstrating the suitability of the ELISA for discriminating these infections. In a further study (3) the Anti-Zika Virus ELISA showed no cross reactivity (100% specificity) in sera from patients with early convalescent DENV infections or suspected secondary DENV infections.
Usefulness of IgA testing
In a recent study investigating the diagnostic usefulness of IgA antibodies, anti-ZIKV antibodies of class IgA, IgM and IgG were analysed at serial time points in patients with confirmed ZIKV infections (4, 5). In two German travellers, IgM was detected early in infection as expected, followed by IgG seroconversion. IgA antibodies showed an initial increase and subsequent decrease. In two Columbian patients with a presumptive background of past flavivirus infection, IgM was persistently below the cut-off in both NS1-based and full virus-based tests, while IgG was already positive within the first week. Analysis of IgA in these patients demonstrated a titre increase, which peaked above the cut-off in week three and four before dropping below the threshold again (Figure 1). Thus, specific IgA may be useful for the diagnosis of acute infections and discrimination from past infections in IgM-negative patients.
Clinical evaluation of IgA ELISA
The NS-1-based Anti-Zika Virus ELISA was used to analyse anti-ZIKV antibodies of class IgA in Columbian patients (n=31) seven to ten days after positive ZIKV RT-PCR. 29 of the patients were positive for anti-ZIKV IgA, representing a sensitivity of 94%. The specificity of the IgA ELISA amounted to 97% in a control panel of German travellers with confirmed DENV infections and 100% in healthy blood donors and patients with other diseases. With the IgA ELISA, as with the IgM and IgG ELISAs, cross reactivity with antibodies against other flaviviruses, including DENV, TBEV, JEV, WNV and YFV, is almost completely avoided.
Differential diagnostics by IIFT
The indirect immunofluorescence test (IIFT) based on virus-infected cells offers an alternative sensitive screening assay for ZIKV antibodies. Automated microscopy and evaluation of results using the EUROPattern system streamlines the procedure. The ZIKV substrate can be combined with other substrates as a BIOCHIP mosaic, enabling potential cross-reactive antibodies or relevant differential diagnostic parameters to be investigated in parallel. In addition to ZIKV, available substrates include DENV (serotypes 1, 2, 3 and 4) and other flaviviruses (e.g. TBEV, YFV and JEV), as well as other arboviruses (e.g. CHIKV). Endpoint titration of the patient serum provides an indication of the virus causing the infection. As cross reactivity is common in patients with secondary flavivirus infections, BIOCHIP flavivirus mosaics are most useful for patients in non-epidemic countries, for example travellers returning from epidemic regions.
Perspectives
The swift development of sensitive and specific tests for ZIKV antibodies and ZIKV RNA has facilitated the diagnosis and surveillance of this rapidly emerging disease. The EUROIMMUN Anti-Zika Virus ELISA based on NS1 antigen is currently the only commercial serological test whose extremely high specificity has been described in various publications. It is, moreover, the first commercial serological ZIKV test to receive CE Mark (Europe; IgA, IgM and IgG) and ANVISA (Brazil; IgM, IgG, soon also IgA) registrations. The assay is fully automatable, making it ideal for high-throughput application in a routine setting. For direct detection of viral RNA, the new EURORealTime Zika virus test provides software-supported test performance and fully automated result evaluation and documentation, in contrast to many manual ZIKV RT-PCR tests. As ZIKV will likely remain a global health challenge in the foreseeable future, state-of-the-art test systems like these are crucial for monitoring the spread, improving diagnosis and elucidating the mechanisms of this challenging emerging disease.
References
1. Steinhagen et al. Euro Surveill. 2016 15;21(50). pii: 30426.
2. Huzly et al. Euro Surveill 2016;21(16):pii=30203.
3. Granger et al. Poster at the 32nd Clinical Virology Symposium (Florida, USA) 2016
4. Steinhagen et al. Poster at the IMED International Meeting on Emerging Infectious Diseases and Surveillance (Vienna, Austria) 2016
5. Steinhagen et al. Poster at the 1st International Conference on Zika Virus (Washington DC, USA) 2017
The author
Jacqueline Gosink, PhD
EUROIMMUN AG, Seekamp 31,
23560 Luebeck, Germany
www.euroimmun.com