Detection of von Hippel–Lindau (VHL) gene copy number variation
Detection of copy number variations (CNV) in the VHL gene is part of the genetic workup of VHL-related tumours. Current methods for CNV determination have complex workflows and limitations. Digital droplet PCR is a promising methodology that could be used for CNV determination. Its advantages include shorter turnaround time, decreased DNA input and superior precision.
by Dragana Milosevic, Dr Stefan K. Grebe, Dr Alicia Algeciras-Schimnich
Background
Von Hippel-Lindau (VHL) disease is an autosomal dominant cancer syndrome with an incidence of approximately 1 in 36,000 live births. It predisposes affected individuals to the development of five main types of neoplasms: retinal angioma (>90% penetrance), cerebellar hemangioblastoma (>80% penetrance), clear-cell renal cell carcinoma (~75% penetrance), spinal hemangioblastoma (~50% penetrance), and pheochromocytoma (~30% penetrance). The disease is caused by mutations or large deletions in the VHL tumour suppressor gene (VHL). The VHL gene is located on chromosome 3p25-26 and encodes a protein that is involved in ubiquitination and degradation of a variety of proteins, most notably hypoxia-inducible factor (HIF) [1]. HIF induces expression of genes that promote cell survival and angiogenesis under conditions of hypoxia. It is believed that diminished HIF degradation due to inactivation of the VHL protein causes the tumours in VHL disease. Tumours form when the remaining intact copy of the VHL gene is somatically inactivated in target tissues.
VHL patients are subdivided in two groups, based on the genotype/phenotype correlations; those at low risk of developing pheochromocytoma are designated type I, whereas those with a high risk of pheochromocytoma (with or without renal cell carcinoma) are classified as type II. Deletions in the VHL gene are more common in type II VHL syndrome [2, 3]. To date, there have been more than 300 germline mutations and large deletions identified in the VHL gene that cause loss of function [2]. Germline loss-of-function point mutations and small deletions or insertions accounts for approximately 70–80% of cases; whereas large germline deletions of one copy of the VHL gene accounts for approximately 20–30% of cases.
VHL genetic testing
The clinical diagnosis of VHL disease is suspected in individuals who present with one or several of the characteristic tumours described above. Molecular genetic testing of VHL is performed to confirm the clinical diagnosis. The genetic testing includes sequencing of the three exons of VHL gene and evaluation of copy number variations (CNV) to assess deletions of large regions of the gene. Historically, detection of these large deletions was done by Southern blot. Today, most clinical laboratories offering CNV determination use multiplex ligation probe amplification (MLPA)-based assays. MLPA is a method based on sequence specific probe hybridization, ligation and PCR amplification and detection of multiple targets with a single set of universal primers. CNVs are detected by comparison of the signal from each target region to control genes and normal control samples.
Although MLPA-based assays are of superior quality and more robust than previous technologies, technical success of MLPA assays is dependent on input of high quantities (at least 400ng of germline DNA) of high quality DNA. Although less labour intensive than Southern blotting, the MLPA work flow is still more complex than PCR-based assays and typically takes two days until completion. Finally, MLPA does not allow for absolute quantification and cannot distinguish copy numbers greater than three with high accuracy.
Digital droplet PCR
Digital droplet PCR (ddPCR) is a methodology that has gained favour as a robust alternative with improved precision to quantitative real-time PCR (qPCR) for DNA quantification. DdPCR also lends itself to exact CNV determination, detection of rare variants, translocations, and/or point mutations (SNP genotyping).
DdPCR is based on traditional PCR amplification and fluorescent probe-based detection methods, but partitions each reaction into 15 000–20 000 nanodroplets. Provided that the starting DNA concentration is not too high, some of these reactions will contain one or more target DNA molecules, whereas others will not contain any. Those with at least one target DNA molecule will yield an amplification product, while those without won’t. Quantification is based on counting the proportion of droplets that show amplification, using a microfluidic counting device. The proportion of reactions with and without amplification obeys Poisson statistics and allows back-calculation of the starting concentration based on the distribution function. When enough droplets are used, copy number ascertainment is of unprecedented accuracy and reproducibility (CVs of 2–10%) [4]. Compared to standard qPCR methods, ddPCR eliminates the need for standard curves and measures both target and reference DNA within the same well. Applications where ddPCR has been used include: rare allele detection in heterogeneous tumours, assessment of tumour burden by analysis of peripheral body fluids (mainly blood), non-invasive prenatal diagnostics, viral load detection, CNV, assays with limited sample material such as single cell gene expression and archival formalin-fixed paraffin-embedded (FFPE) samples, DNA quality control tests before sequencing, and validation of low frequency mutations identified by sequencing.
Recently, ddPCR has become commercially available in a format that allows for rapid microfluidic analysis of thousands of droplets per sample making it practical for routine use in clinical laboratories. A recent study on the analytical performance of ddPCR has shown greater precision (CVs decreased by 37–86%) and improved day-to-day reproducibility and comparable sensitivity to real-time PCR for absolute quantification of microRNAs [5]. A study that evaluated the use of ddPCR to detect BCR-ABL1 fusion transcripts demonstrated that ddPCR is able to achieve lower limit of detection and quantification than currently used in quantitative PCR methods [6].
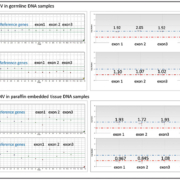
Our group has evaluated ddPCR for VHL CNV and shown improved performance compared to MLPA [7]. The method showed 100% concordance with the MLPA method and 100% self-concordance within and between runs. The method showed reproducible results with DNA inputs as low as 10 ng, a 40-fold DNA-input reduction compared with MLPA. Because of this advantage, difficult specimen types, such as archival FFPE specimens, are now capable of being characterized for VHL CNVs, a feat previously impossible by MPLA, because of the often poor DNA quality of such samples (Fig. 1). Additionally, same-day results are available with the ddPCR method, reducing the total run-time from 48 hours for the MLPA method to 3 hours.
One limitation of current ddPCR platforms is the limited ability for multiplexing. For example the Bio-Rad’s ddPCR system can detect only two colours (FAM and HEX), limiting the number of genes that could be evaluated simultaneously in a single reaction. Development of platforms that allow greater multiplexing should, therefore, further facilitate the adaptation of this technology in clinical laboratories.
Conclusions
Improvement of current methods for VHL CNV testing is desired to obtained accurate and cost-effective results in clinical laboratories. Currently used methods are still labour intensive and not suitable for rapid turnaround time. DdPCR is an elegant adaptation of the current quantitative PCR format and has the potential to be applied widely in clinical laboratories. For VHL CNV, ddPCR provides a greatly improved turnaround time and requires only minimal nucleic acid input that does not have to be of the highest quality. With no need for standard curves or controls, ddPCR overcomes the issues associated with traditional qPCR, while increasing both robustness (superior sensitivity, specificity, and precision) and utility in other specimen types such as paraffin-embedded tissue, circulating cell-free DNA, circulating tumour cells, and microRNA detection [8–10].
References
1. Richards FM. Molecular pathology of von Hippel–Lindau disease and the VHL tumor suppressor gene. Expert Rev Mol Med. 2001; 3: 1–27. DOI: http://dx.doi.org/10.1017/S1462399401002654.
2. Maher ER, Kaelin WG Jr. von Hippel-Lindau disease. Medicine 1997; 76: 381–391.
3. Hes FJ, Höppener JW, Lips CJ. Clinical review 155: pheochromocytoma in von Hippel-Lindau disease. J Clin Endocrinol Metab. 2003; 88: 969–974.
4. Pohl G, Shih IeM. Principle and applications of digital PCR. Expert Mol Rev Diagn. 2004; 4: 41–47.
5. Hindson CM, Chevillet JR, Briggs HA, Gallichotte EN, Ruf IK, Vessella RL, Tewari M. Absolute quantification by droplet digital PCR versus analog real-time PCR. Nat Methods 2013; 10: 1003–1005.
6. Jennings LJ, George D, Czech J, Yu M, Joseph L. Detection and quantification of BCR-ABL1 fusion transcripts by droplet digital PCR. J Mol Diagn. 2014; 16(2): 174–179.
7. Milosevic D, Grebe SK, Algeciras-Schimnich A. Detection of Von Hippel-Lindau (VHL) gene copy number variations using digital droplet PCR. Clin Chem. 2014; 60(10S): S194.
8. Wang J, Ramakrishnan R, Tang Z, Fan W, Kluge A, Dowlati A, Jones RC, Ma PC. Quantifying EGFR alterations in the lung cancer genome with nanofluidic digital pcr arrays. Clin. Chem. 2010; 56: 623–632.
9. Lo YM, Lun FM, Chan KC, Tsui NB, Chong KC, Lau TK, Leung TY, Zee BC, Cantor CR, Chiu RW. Digital PCR for the molecular detection of fetal chromosomal aneuploidy. Proc. Natl Acad Sci U S A 2007; 104: 13116–13121.
10. Pinheiro LB, Coleman VA, Hindson CM, Herrmann J, Hindson BJ, Bhat S, Emslie KR. Evaluation of a droplet digital polymerase chain reaction format for DNA copy number quantification. Anal Chem. 2012; 84: 1003–1011.
The authors
Dragana Milosevic MS; Stefan K. Grebe MD, PhD; Alicia Algeciras-Schimnich* PhD
Department of Laboratory Medicine and Pathology, Rochester, MN, USA
*Corresponding author
E-mail: Algeciras.Alicia@mayo.edu