Digital morphology
What advances mean for modern laboratories, and why continued innovation is imperative
by Dr Gina Zini
Advances in technology mean that moving from microscopes to digital alternatives can now be achieved without sacrificing image quality. But this isn’t about the technology. Dr Gina Zini reflects on the key benefits of digital developments, including better quality screening, saved time for busy morphologists, better clinical collaboration, improved training, and sharing expertise with developing countries.
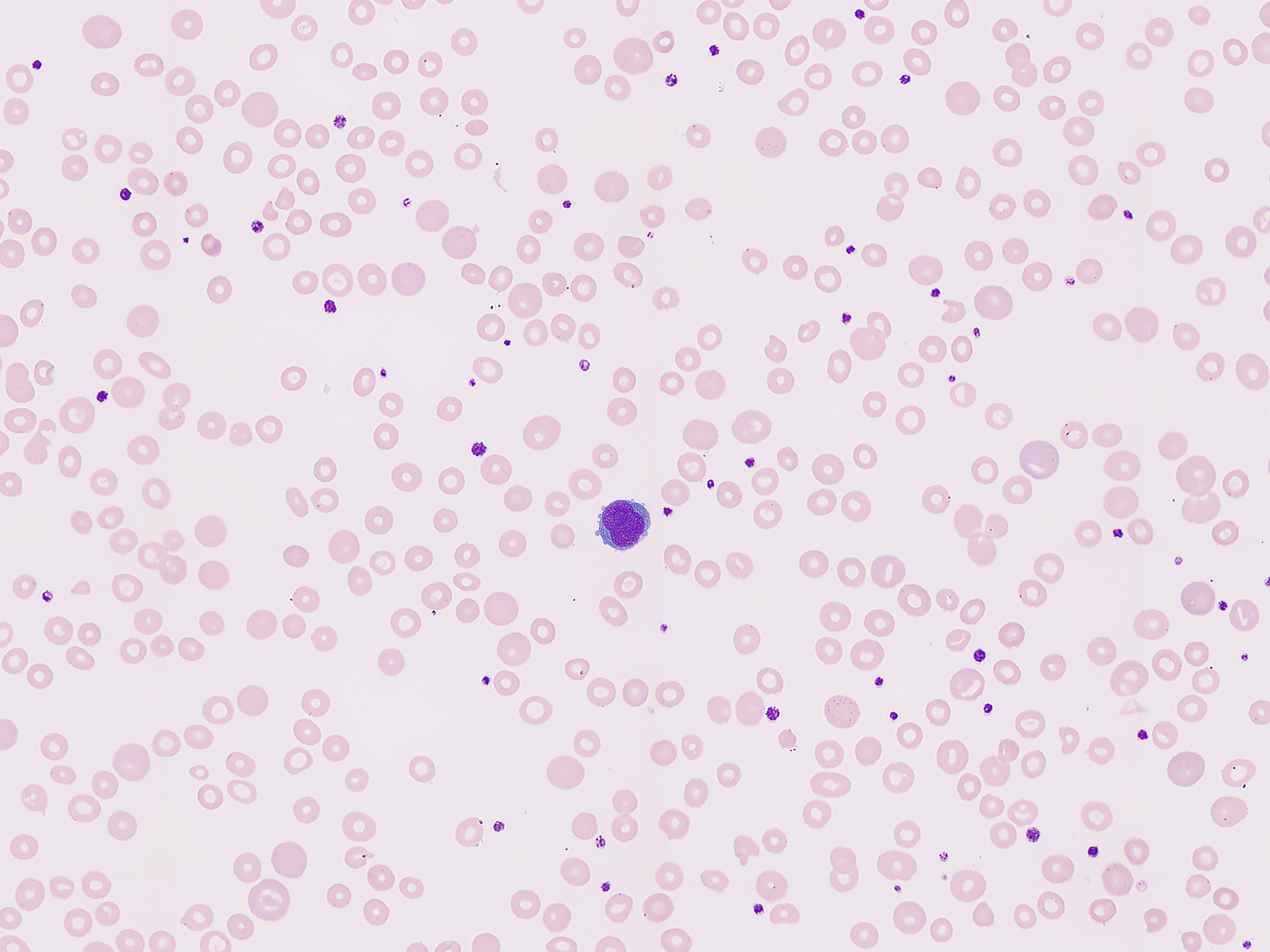
Image of blood cells (Mindray)
When I started my career as a hematologist, examining a blood smear meant looking through a microscope. This was the standard throughout the world. Advances in technology have since led to the development and deployment of new approaches in many countries. In particular, digital tools have played increasingly supportive roles, often replacing or reducing the need for many manually intensive tasks in laboratories.
The potential for digital, particularly in areas such as hematology and morphology where I specialize, is now expanding further as more sophisticated technologies continue to be introduced. The aims are to improve efficiency, quality and collaboration in an environment where scarce professionals are in high demand.
What have laboratories been achieving in digital morphology?
Digital morphology analysers have been adopted in many parts of the world as one means to improve efficiency in the laboratory, as well as the quality of reporting in screening programmes. Such devices have helped to automate parts of the screening process, analysing large quantities of blood smears to help to identify abnormalities. Technology certainly hasn’t replaced the role of hematologists. Human interaction still plays a very substantial role in screening programmes, with hematologists intervening when cases are flagged, and validating significant samples in an ongoing process to help to ensure accuracy and reliability.
Modern digital morphology analysers have allowed professionals in the laboratory to view, validate and report from images on-screen, enabling them to more easily find cells and areas of concern than if they were looking through a microscope.
Recalling images on the screen, compared to searching for the cells on a slide under the microscope also represents a great advantage in terms of both the necessary time and effort for busy laboratory teams. The on-screen availability of digital cellular images has facilitated intra-laboratory, and sometimes inter-laboratory, consensus and harmonization, with images easily accessible to and searchable by teams who are onsite and those working remotely. Access to digital images can, for example, allow staff
to share images with other colleagues for expert opinion, often even if that colleague is working at home.
Additional gains have also been presented by digital technologies.
A blood smear can sometimes provide the primary – or the only – evidence of a specific diagnosis, such as myelodysplastic syndrome, leukemia, lymphoma or hemolytic anemia. Such blood smears may need to be stored over the long term, which is more easily achievable with digital morphology images.
And for some abnormal parameters provided by the automated analysers, such as red blood cell abnormal parameters, accuracy can be higher and less subjective than grading using microscopy. This can help staff to deliver important reports after conducting only a rapid qualitative assessment of the smear.
In short, digital morphology has been delivering significant advantages in laboratories seeking to improve turnaround times, quality and costs – helping to alleviate some of the fatigue and workload challenges associated with optical morphology, and improving the opportunity for repeatability and the easier management of cell images.
Why continued innovation is urgent and imperative, and where it is already happening
The need for highly capable digital tools has now intensified. Their role is transforming from supporting for the efficient business of the laboratory, to one of an urgent necessity for screening services under pressure. There is now a crisis of human resources in fields such as hematology and morphological analysis, fuelled by the availability of fewer experts each year, who continue to manage an ever-increasing workload to support preventative healthcare. The situation now requires even better technologies to augment capacity.
Digital morphology analysers are an important example of technology that has evolved to support this, and where innovation continues. Historically, although helpful, older devices have presented some limitations. Many of these devices have worked by rebuilding an image, rather than displaying a true image of the smear. This means that images appear differently to those seen on glass slides through optical microscopy. Consequently, laboratory teams reviewing an image have needed additional training to be taught how to interpret images and recognize areas of concern from images produced by specific devices, in order to be able to report in an effective and consistent manner.
This is now changing, with new digital technologies able to reproduce precisely what is seen under a microscope. This was shown to be the case in a performance analysis that I recently contributed to around the MC-80 automated digital cell morphology analyser (Mindray).
Blood samples examined in the analysis demonstrated excellent reproducibility of cell images, which were a perfect match to those seen under a microscope. I knew nothing of the cases before the study, but I could immediately check and validate images on the screen. And this is made possible by the fact that for each sample evaluated, within the time of a click of the mouse, two hundred pre-classified images of nucleated cells appear on the screen with morphological details identical to those observed under an optical microscope.
There is therefore no need for any additional time to locate the slide, position it on the microscope table, search for the observation and counting area, focus with objectives at different magnifications including the addition of oil, as occurs when working with an optical microscope. Time spent is dedicated to observing the cells and confirming their pre-classification or moving to a different subclass, when necessary; however, after the movement, the system will automatically regenerate the percentages of the differential.
To evaluate this series of samples, which were all pathological due to quantitative and/or qualitative anomalies, I spent an average of one to two minutes per sample for screen validation. It took an average of six to eight minutes to produce the reference differential under an optical microscope, evaluating in both cases at least 200 nucleated elements.
This ability to create images that are indistinguishable from the microscopy view is state of the art. Efficiency gains and support for better quality reporting are benefits that speak for themselves in the context of stretched resources. But it also opens up new possibilities around training and collaboration. Digital morphology can in general reduce the time it takes to train a good morphologist. But it is also worth bearing in mind that despite growing adoption, only around 37% of the world uses digital morphology. Many developing countries, for example, still rely on microscopy as their primary tools.
Having access to technologies that can reproduce an image as seen under a microscope, opens immediate opportunities for laboratories using digital morphology to share recognizable images for training purposes with professionals and trainees in countries where the technology is not yet widely used. And if we can increase adoption of digital morphology that produces images familiar to those who use microscopy, opportunities to improve screening, reduce false negatives, and extend the many other benefits that digital morphology can bring could be realized.
Image of blood cells (Mindray)
The author
Assoc. Prof. Gina Zini PhD
Università Cattolica del Sacro Cuore, Rome, Italy Fondazione Policlinico Universitario A. Gemelli IRCCS, Rome, Italy
Email: gina.zini@unicatt.it