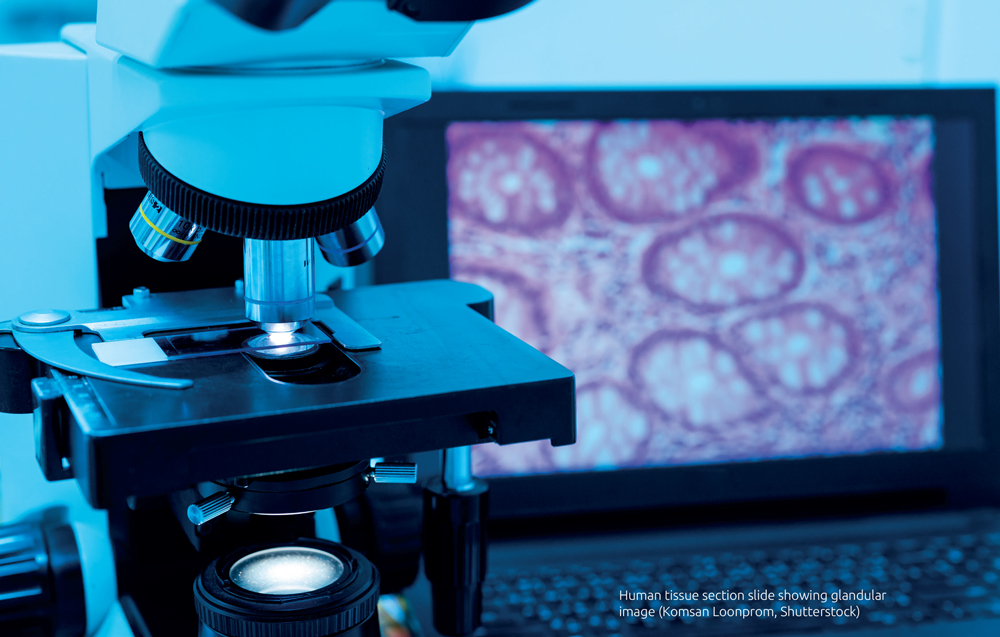
Digital pathology in the clinical lab
Histopathology is a crucial stage in the diagnosis of many diseases, particularly cancer staging and therapy management. Current challenges facing pathology are a shortage of pathologists and an increasing workload resulting from expanded screening programmes, increased test complexity and an increasing elderly population. Leeds Teaching Hospitals NHS Trust, UK, has a fully subspecialized diagnostic histopathology department that is home to the world’s most long established digital pathology project. CLI caught up with Dr Bethany Williams, Fellow in Digital Pathology, at Leeds to find out more about the process of going digital and what the benefits and challenges are.
Arguably, the use of microscopy in disease diagnosis began in the late 1600s when Anton van Leeuwenhoek developed a simple light microscope that led to the discovery of bacteria. Can you describe what histopathology is?
Clinical histopathology is a medical specialty concerned with human tissue diagnosis. Human tissue specimens including biopsies and surgical specimens arrive at the laboratory, where selected pieces of tissue are processed and mounted on glass slides, so they can be viewed by highly specialized doctors, pathologists using a microscope. The pathologist evaluates the tissue to provide vital diagnostic and prognostic information to the surgeon, referring doctor, and patient. For cancer specimens, pathologists use the microscope to provide vital information that directs patient treatment, including the definitive diagnosis, the cancer subtype, grade and stage.
Leeds Hospital is currently changing over to the use of digital pathology – what is digital pathology and what does this entail?
Digital pathology is a technology with the potential to revolutionize the way in which cancer diagnostic services are delivered. In digital pathology, the glass slides created in the laboratory are scanned using a desktop scanner containing a quality microscope lens, which captures a high resolution digital image of the slide. This digital slide can be accessed and viewed by a pathologist using specialized microscope software, on a computer screen. Histopathology services are under increasing pressure and face the challenges of increasing workloads (in terms of the number and complexity of cancer cases received each year), and dwindling workforce. The flexibility and transferability of the digital slide medium compared with standard glass slides and microscopes can help to create robust, efficient cancer diagnostic networks.
Leeds has a track record in digital pathology research and innovation, but it was not until 2015 that we felt the technology had reached a stage where we could trial digital pathology in the clinical for primary diagnosis. When we made the decision to ‘go digital’ in our NHS laboratory, we wanted to be sure our actions were evidence based, and that patient safety and professionalism were our first concerns. At this time, we formed a collaborative research partnership with Leica Biosystems, which allowed us to investigate the safety of digital pathology diagnosis, and develop training and validation protocols for doctors converting to digital pathology practice.
What are the benefits of digital pathology?
The principal benefits of digital pathology include improvements in patient safety, service efficiency, workforce factors and service quality. Firstly, the digital slide is not subject to the same risks as the glass slide – it does not fade, degrade, get lost or broken. Use of digital slides as part of a paperless pathology reporting system also lessens the risk of patient identification errors. Digital slides are flexible and easily transferable, enabling innovative, more efficient workload allocation to pathologists, faster case tracing and retrieval, and more rapid access to second opinion. This transferability allows more effective information sharing and collaboration between pathologists, whether they are in the same hospital, or further afield. This aspect of the technology could also help with recruitment and retention issues in pathology, providing the possibility for pathologists to work remotely and flexibly, and improving training and teaching opportunities.
What are the challenges/limitations of digital pathology?
Deploying a digital pathology system is a major change management project, requiring changes in the laboratory and the diagnostician’s office. A full departmental deployment can be costly, requiring new hardware, software, and infrastructure upgrades.
Can you envisage a time when analysis of slides and diagnosis by computer algorithms replaces the use of the human pathologist?
The human pathologist is a medically trained doctor, with many years of experience in human tissue diagnostics, able to detect subtle ‘picture’ clues in pathology slides and integrate these with a patient’s medical history to provide a diagnosis, and information which helps fellow doctors and surgeons plan treatments with patients. Current artificial intelligence applications can assist the pathologists by helping to answer narrow, clearly defined questions such as: Is this immunostain positive? and Does this lymph node contain abnormal cells? However, a trained medical professional will still be required for the bulk of diagnostic tasks for the foreseeable future.
I see a future (on a very close horizon!) where hybrid diagnosis becomes standard. The pathologist allows the computer to take over some of the more onerous, time consuming counting and quantifying tasks (e.g. counting mitotic figures that reflect how aggressive a tumour is, measuring the volume of tumours) while the pathologist can devote more time to the more intellectual challenges of diagnosis. This type of model could improve the efficiency of cancer diagnosis, and help future-proof a vital, stretched medical specialty.
The expert
Bethany J. Williams MBBS, PhD; Digital Pathology Fellow
National Pathology Imaging Co-Operative, St James University Hospital, Leeds, UK
E-mail: bethany.williams2@nhs.net