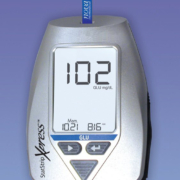
Glucose meter system receives FDA clearance for use with critically ill patients
Last May, the U.S. Food and Drug Administration (FDA) cleared Nova Biomedical’s StatStrip Xpress Glucose Hospital Meter System for use throughout all hospital and all professional healthcare settings, including critically ill patients. StatStrip Glucose and StatStrip Xpress Glucose are now the only two hospital blood glucose meters to be cleared by the FDA for use with critically ill patients. Use of all other glucose meters with critically ill patients is considered off-label by the FDA and high complexity testing under the Clinical Laboratory Improvement Amendments. High complexity testing requirements are so stringent that to use a glucose meter other than StatStrip Glucose and StatStrip Xpress Glucose with critically ill patients is not a practical alternative. StatStrip Xpress Glucose utilizes the same test strip measurement technology as StatStrip Glucose, which was cleared in 2014 after an extensive, four-year study conducted at five major university medical centres. The study included 1,698 critically ill patients with over 257 medical condition subcategories as designated by the World Health Organization. Over 8,000 medications were investigated for potential interference to StatStrip Glucose measuring technology. StatStrip Glucose demonstrated excellent agreement compared to central laboratory reference methods and no clinical interferences were found. In addition to the study submitted to the FDA, 138 other independent studies over the last eight years—including 53 critical care studies—have found no clinically significant interferences for StatStrip Glucose measuring technology.