Insight into monocyte distribution width for sepsis diagnosis and COVID-19 prognosis
Sepsis requires fast treatment for the best outcome, but is notoriously difficult to diagnose. Monocyte distribution width (MDW) is a parameter that gaining traction in the diagnosis of sepsis and may also be useful in predicting severe outcomes from COVID-19 infection. CLI caught up with Lorenzo Maggia, Product and Scientific Senior Manager at Beckman Coulter Europe, and Dr Giovanni Riva, Senior ED Physician at Modena Hospital, to discover more about the benefits of assessing MDW.
In the October 2020 issue of CLI we published the article ‘Monocyte distribution width: new biomarker for sepsis diagnosis’, but could you begin with a brief recap of what sepsis is, how it arises and why a timely diagnosis (or rule-out) is so important?
Lorenzo Maggia (LM): Sepsis represents a dysregulated immune response to infection that leads to organ dysfunction [1]. This definition, while the most appropriate, though driest, definition of sepsis, hides the complexity of the subject matter. Sepsis is not a specific illness but rather a syndrome encompassing a still-uncertain pathobiology. Sepsis is identified by a constellation of clinical signs and symptoms; diagnosis is still very subjective, particularly in the early stages. No single test or scoring system can perfectly identify septic patients [2,3], and it is difficult to distinguish sepsis from non-infectious causes of acute illness because these patients often share common clinical signs and symptoms.
All of the above factors lead to potential delays in one of the most critical parts of treatment – the administration of antibiotics. Current guidelines emphasize the benefits of early detection and treatments.
The literature also demonstrates that every hour for which the administration of antibiotics is delayed for patients with severe sepsis and septic shock leads to an increased mortality of 7.6% [4]. This is very impressive. Another recent publication in Chest, based on a review of data relating to over 10 000 Emergency Department (ED) patients, demonstrated that each additional hour – from arrival at the ED to antibiotic initiation – meant a 10% (95% CI, 5–14; P<0.001) increase in 1-year mortality [5].
Therefore, Beckman Coulter worked on releasing monocyte distribution width (MDW), [a U.S. Federal Drug Administration (FDA in the USA) and European conformity (CE) cleared in vitro diagnostic (IVD) test] that, in conjunction with other clinical para-
meters, allows clinicians to escalate and de-escalate care in patients with suspected infections.
What is MDW and how is it useful in the diagnosis of sepsis? How does it compare to other biomarkers of sepsis?
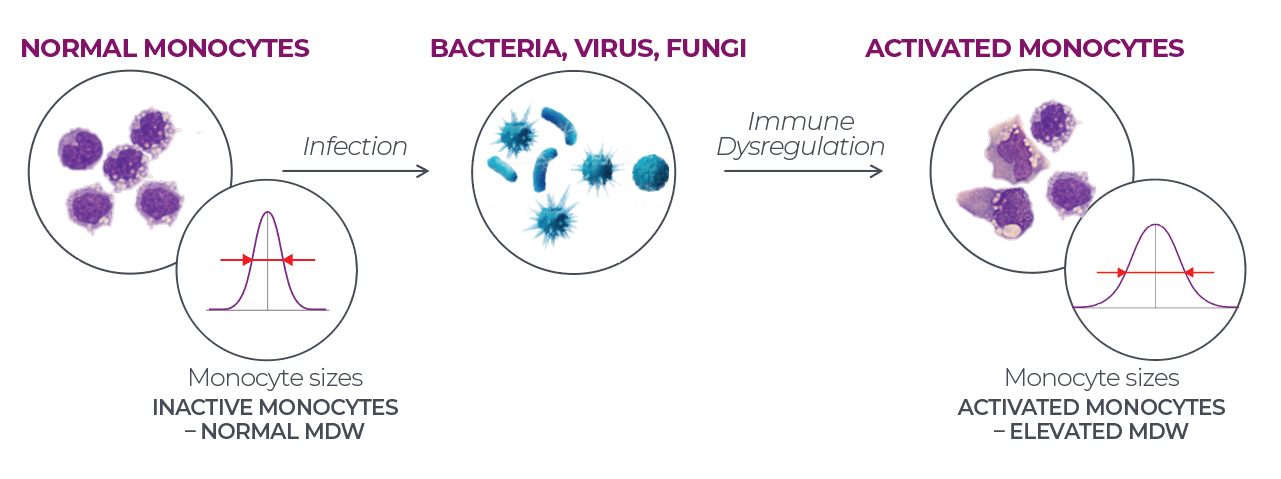
LM: MDW is a hematologic parameter measured as part of the complete blood count with differential (CBC-DIFF). It describes the size distribution of circulating monocytes, a subset of cells that are considered key mediators of the life-threatening hyperinflammatory disorder characterizing sepsis (Fig. 1).
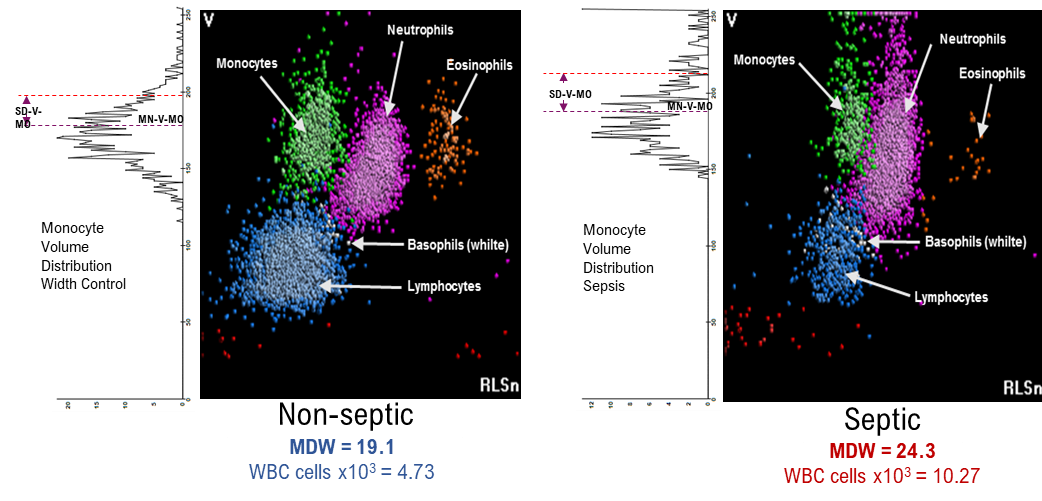
Activation of monocytes in response to infection results in some monocytes getting bigger; the DxH900 analyser is able to precisely measure these changes in volume (Fig. 2).
Clinical validity and performance of MDW have been confirmed by multiple studies and clarify the role of this test as helpful in identifying patients who have developed sepsis or severe infection [6,7].
A very recent publication of European clinical trials has demon-strated that MDW performance for sepsis detection is comparable with other biomarkers, such as procalcitonin and C-reactive protein (CRP) [8]. The area under the curve (AUC) values (95% confidence interval) for the diagnosis of Sepsis-2 were 0.81 (0.78–0.84) and 0.86 (0.84–0.88) for MDW and MDW combined with white blood cell (WBC) count,, respectively. For Sepsis-3, MDW performance was 0.82 [0.79–0.85]. The AUC for sepsis-2 detection using MDW combined with WBC was similar to CRP alone [0.85 (0.83–0.87)] and exceeded that of PCT [0.78 (0.75–0.81)]. It is important to note that abnormal MDW and WBC at the time of ED admission may alert to potential severe infection/sepsis even in circumstances of low clinical suspicion, as AUC in a subgroup of patients with low sepsis pre-test probability was 0.90 (0.84–0.95). The authors suggested that the use of MDW as a systematic screening test, when used with the quick Sepsis Related Organ Failure Assessment (qSOFA) score, improves the accuracy of sepsis diagnosis in the ED.
MDW has the diagnostic accuracy to detect sepsis, particularly when assessed in patients with lower pre-test sepsis probability. The majority of these publications suggest the use of MDW as a routine screening test, used along with Systemic Inflammatory Response Syndrome (SIRS) criteria and the qSOFA score, to improve the accuracy of sepsis diagnosis in the ED.
How easy could implementation be in hospitals, and what would be the impact of this implementation?
LM: MDW is part of a standard CBC-DIFF test, one of the most common requests already performed in hospitals; I think implementation is just a matter of updating a shared standard operating procedure (SOP) that integrates this new test with a broader decision chart. MDW could improve decision-making during the early management of sepsis infections. This is a significant difference from other biomarkers that must be specifically requested.
Across Europe, more and more sites are putting our MDW in action. Joint efforts made due to laboratory and ED implementation are already increasing the collection of evidence of the impact of MDW and beyond. As the COVID-19 pandemic took hold, our customers started to report cases of a connection between MDW and COVID-19.
MDW can help detect patients infected with the Covid-19 virus, presenting with good sensitivity (87%). Even more interestingly, abnormal MDW increased the odds of identifying more severe cases of the COVID-19 disease. MDW may have implications in identifying patients who need more immediate attention.
As we have learnt during the course of the COVID-19 pandemic, severe SARS-CoV-2 infection can trigger a cytokine storm. Can MDW be of use in diagnosing COVID-19 and predicting severe illness?
Giovanni Riva (GR): From the advent of this pandemic, it was soon evident that monocyte/macrophage activation plays a key role in the systemic hyper-inflammatory disorder characterizing severe COVID-19. Intriguingly, by providing a conceptual link between the diagnosis of sepsis and COVID-19, MDW can support the notion that severe COVID-19 may constitute a (new) kind of viral sepsis. In this view, by considering both the biological rationale and clinical data reported so far, MDW can present a useful hematological (cell-based) parameter directly related to the ‘abnormal monocyte activation’ occurring in life-threatening (dis)inflammatory diseases, such as sepsis and COVID-19. Accordingly, in a recent paper published in EBioMedicine [9], we proposed a working model for the prognostic use of MDW in this setting. More specifically: high MDW values (>26) can help identify COVID-19 patients with severe cytokine storm, at high risk of death; patients with intermediate MDW values (ranging from 20 to 26) usually show mild-to-moderate COVID-19 (i.e. persistent disease, but not so critical for the time being); whereas low MDW values (<20) are shown by patients heading towards full recovery, as well as, most probably, by asymptomatic COVID-19 cases with low/absent systemic inflammation.
How might MDW help with the clinical management of COVID-19 patients?
GR: In our recently published study in Scientific Reports [10], we showed that MDW can correlate with COVID-19-related systemic inflammation, disease severity and patient outcome and could, therefore, serve as a prognostic biomarker in hospitalized COVID-19 patients. In particular, we found that high MDW values identified using 26.4 as the AUC-receiver operating characteristic (ROC) threshold were associated with elevated mortality risk (about one third) in our series of 87 COVID-19 patients. Possibly in line with this finding, a mean MDW value of 28.8 was found in a series of 41 very critical COVID-19 patients tested once for MDW at ED admission. In contrast, a mean value of 25.4 was observed in patients with milder symptoms, thus supporting the idea that severe clinical conditions can be linked to higher MDW values. However, MDW is a recent biomarker, and further validation studies are now required to define optimal cut-off values for prognostic MDW testing.
Are there any other scenarios where MDW analysis might be of use?
LM: MDW is actually intended to be used only on adult patients entering the ED, and both clinical trials were based on this population. It’s a very restricted population but one with an immensely diverse causality; this means that there are potentially multiple scenarios where MDW can be applied and with a simplistic approach, we can define these scenarios as all hospitalized patients.
The first focus was placed on the ICU, about which there is an increasing number of publications. For example, Piva et al. [11] and Agnello et al. [12] confirmed the possible application of MDW in ICU in two different university hospitals in Italy. In both studies, MDW was statistically higher in those with sepsis and septic shock compared to those without sepsis (P<0.001), whereas no statistical differences were found between sepsis and septic shock patients.
However, although these results are encouraging, more clinical studies are needed to verify the findings in different populations, setting appropriate cut-off values and augmenting the
areas of its application
Figure 1. Infection results in the activation of circulating monocytes, causing in an increase in their size, which is reflected in an increased monocyte distribution width
Figure 2. Precisely measuring the volume of monocytes enables the differentiation of infection-induced sepsis or non-septic illness
The interviewees
Dr Giovanni Riva1,2 MD, PhD, Senior Physician and Lorenzo Maggia3, Product and Scientific Senior Manager at Beckman Coulter Europe.
1S.S.D. Ematologia Diagnostica e Genomica Clinica, Dip. Interaziendale ad Attività Integrata Medicina di Laboratorio e Anatomia Patologica, University of Modena, Modena, Italy
2Ambulatorio Ematologia, Dip. Cure Primarie, AUSL di Modena, Modena, Italy
3Beckman Coulter Europe, 20060 Cassina de’ Pecchi, Milan, Italy
For further information visit Beckman Coulter:
https://www.beckmancoulter.com/en/products/hematology/
early-sepsis-indicator
References
- Singer M, Deutschman CS, Seymour CW et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA 2016;315(8):801–810 (https://jamanetwork.com/journals/jama/fullarticle/2492881).
- Seymour CW, Liu VX, Iwashyna TJ et al. Assessment of clinical criteria for sepsis: for the Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA 2016;315(8):762–774. doi: 10.1001/jama.2016.0288 (https://jamanetwork.com/journals/jama/fullarticle/2492875).
- Ljungström L, Pernestig AK, Jacobsson G et al. Diagnostic accuracy of procalcitonin, neutrophil-lymphocyte count ratio, C-reactive protein, and lactate in patients with suspected bacterial sepsis. PLoS One 2017;12(7):e0181704 (https://doi.org/10.1371/journal.pone.0181704).
- Kumar A, Roberts D, Wood KE et al. Duration of hypotension before initiation of effective antimicrobial therapy is the critical determinant of survival in human septic shock. Crit Care Med 2006;34(6):1589–1596.
- Peltan ID, Brown SM, Bledsoe JR et al. ED door-to-antibiotic time and long-term mortality in sepsis. Chest 2019;155(5):938–946 (https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6533450/).
- Crouser ED, Parrillo JE, Seymour CW et al. Improved early detection of sepsis in the ED with a novel monocyte distribution width biomarker. Chest 2017;152:518–526 (https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6026271/).
- Crouser ED, Parrillo JE, Martin GS et al. Monocyte distribution width enhances early sepsis detection in the emergency department beyond SIRS and qSOFA. J Intensive Care 2020;8:33 (https://jintensivecare.biomedcentral.com/articles/10.1186/s40560-020-00446-3).
- Hausfater P, Robert Boter N, Morales Indiano C et al. Monocyte distribution width (MDW) performance as an early sepsis indicator in the emergency department: comparison with CRP and procalcitonin in a multicenter international European prospective study. Crit Care 2021;25(1):227 (https://ccforum.biomedcentral.com/articles/10.1186/s13054-021-03622-5).
- Riva G, Nasillo V, Luppi M et al. Linking COVID-19, monocyte activation and sepsis: MDW, a novel biomarker from cytometry. EBioMedicine 2022;75:103754 (https://doi.org/10.1016/j.ebiom.2021.103754).
- Riva G, Castellano S, Nasillo V et al. Monocyte distribution width (MDW) as novel inflammatory marker with prognostic significance in COVID-19 patients. Sci Rep 2021;11(1):12716
(https://www.nature.com/articles/s41598-021-92236-6) - Piva E, Zuin J, Pelloso M et al. Monocyte distribution width (MDW) parameter as a sepsis indicator in intensive care units. Clin Chem Lab Med 2021;59(7):1307–1314 (https://doi.org/10.1515/cclm-2021-0192).
- Agnello L, Sasso BL, Giglio RV et al. Monocyte distribution width as a biomarker of sepsis in the intensive care unit: a pilot study. Ann Clin Biochem 2021;58:70–73 (https://doi.org/10.1177/0004563220970447).