Introducing serum apolipoprotein profiling for enabling precision cardiology: its time has come
To assess the risk of cardiovascular disease (CVD), 10 – year risk scores are calculated based on four serum lipids (total cholesterol, LDL-C, HDL-C and total triglycerides) and other (un)modifiable risk factors. Recently we proposed an apolipoprotein (apo) panel that provides more granular information regarding the specific type of lipid disorder. This extended information fits the concept of precision medicine and provides support to the unmet clinical need for stratified cardiovascular medicine.
By Dr L. R. Ruhaak, Prof. A. van der Laarse
and Prof. C..M. Cobbaert
Introduction
In primary and secondary prevention of cardiovascular disease (CVD) the presence of risk factors such as dyslipidemia, diabetes mellitus (DM), abdominal obesity, among others, are diagnosed and treated. A lipid panel consisting of total cholesterol (TC), total triglycerides (TG), high-density lipo-protein-cholesterol (HDL-C) and (calculated) low-density lipoprotein-cholesterol (LDL-C) is currently measured for diagnosis and monitoring of dyslipidemia, but this lipid panel does not allow precise typing of dyslipidemias, nor does it highlight under-lying pathways and/or lipidemic factors that are highly atherogenic. This results in blockbuster therapies (using mostly statins) and substantial residual cardiovascular risk, even if the lipid profile seems normal. Therefore, there is an unmet clinical need [1] for the development of better markers for CVD risk assessment. To give emphasis to proper diagnosis of dyslipoproteinemias and to reduce the number of patients with significant residual cardiovascular risk despite therapy, we should shift from ‘reactive medicine’ that generally treats an average patient with an average drug dose (for instance, treatment of a hypercholesterolemic patient by a statin) to ‘pro-active P4 medicine’ (predictive, preventive, personalized and participatory) for a personalized medicine approach using markers that are associated with pathophysiological processes and allow for better understanding and management of interindividual heterogeneity [2, 3]. One such approach could be the quantitation of the functional apolipoproteins (apos) that are associated with lipid metabolism. Indeed, for decades, Sniderman et al. pioneered the development of apolipo-protein B (apoB) testing, beyond TC and TG tests, to improve the diagnosis and treatment of dyslipoproteinemias [4]. To facilitate diagnosis and treatment of lipid disorders, we developed a broader, multiplexed panel of clinically relevant apos [5]. Below, we summarize the characteristics of the apos in our multiplex panel as well as the evidence for their role in dyslipidemias.
Biochemistry and pathobiology of apolipoproteins
ApoB
Human apoB100 is a 4536-amino acid secretory glycoprotein. A single molecule is present in each very-low-density lipoprotein (VLDL) particle when secreted by the liver. After entering circulation, lipoprotein lipase (LPL) and hepatic lipase (HL) are responsible for lipolysis of TG in the VLDL particle. The resulting VLDL-remnants are either cleared by the liver or converted to LDL by further lipolysis of TG. LDL-C is a strong risk factor for atheroscle-rotic diseases such as CVD, and a particular family of drugs, statins, reduce the LDL-C levels in blood and decrease the risk of CVD in both primary and secondary prevention programmes. Subsequently, other types of LDL-C lowering drugs were developed, such as ezetimibe and PCSK9-inhibitors. Measurement of serum apoB concentration may detect an increased number of LDL particles often missed with LDL-C alone, as occurs in the atherogenic condition of small dense LDL. This condition is commonly not associated with elevated LDL-C levels but with an increased number of small LDL particles leading to elevated concentrations of apoB [2].
Apart from VLDL, intermediate-density lipoprotein (IDL) and LDL, a fourth lipoprotein type, lipoprotein(a) [Lp(a)], carries apoB. Lp(a) is composed of an apoB-containing LDL-like particle, covalently linked to the plasminogen-like glycoprotein apo(a). Each particle of LDL, VLDL, VLDL-remnants and Lp(a) contains one apoB molecule, thereby making the plasma apoB concentration an important risk factor of CVD, and a target in patients with hypertriglyceridemia, type 2 DM, obesity, or metabolic syndrome [2]. Abundancy of remnant particles usually presents with hypertriglyceridemia and is an important atherogenic factor [6].
In the intestines, a truncated form of apoB is formed corresponding to its N-terminal 48 % (apoB48) which resides in the chylo-microns (CMs) formed in the intestines after having a meal. These TG-rich and cholesterol-poor particles undergo lipolysis of TG in the circulation and their remnants are either taken up by the liver or accumulate as CM-remnants in the blood.
ApoCI
ApoCI, a polypeptide of 57 amino acids, is produced by the liver and is a constituent of VLDL and HDL. ApoCI is an inhibitor of LPL activity, an inhibitor of the cholesteryl ester transfer protein (CETP) and an activator of phosphatidylcholine-sterol acyl transferase [or lecithin-cholesterol acyltransferase (LCAT)] leading to elevated levels of HDL-C.
ApoCII
ApoCII is a polypeptide of 79 amino acids and an essential cofactor of LPL. Individuals who are completely deficient in apoCII have chylomicronemia and elevated plasma TG concentrations.
ApoCIII
ApoCIII is a 79-amino acid polypeptide that is produced mainly in the liver and to a lesser extent in the intestines. ApoCIII is a significant component of TG-rich lipoproteins (TRLs), particularly remnants. Elevated levels of apoCIII are associated with augmented production and retarded clearance of TRLs, and correlated with plasma TG and VLDL-TG. The development of antisense oligonucleotide therapy directed towards the hepatic mRNA of apoCIII, such as volanesorsen, is promising, as dose-dependent reductions in TG levels of up to 80.% are attainable [7]. Individuals with apoCIII deficiency or loss-of-function mutation of APOC3 have reduced risk of CVD, as these conditions promote cardioprotection [8, 9].
ApoE
ApoE, a 299-amino acid polypeptide, plays a key role in the receptor-mediated uptake of apoB100-containing lipoproteins including remnant lipoproteins. Plasma apoE is mainly synthesized by the liver. ApoE has polymorphisms indicated by apoE2, apoE3 and apoE4. ApoE4 is associated with higher plasma cholesterol than apoE3 and is an independent risk factor for CVD. Homozygotes of apoE2 have lower plasma cholesterol than homozygotes of apoE3 which is proposed to have low risk of CVD. However, due to secondary precipitating factors, individuals with apoE2/E2 may develop type III hyper-lipoproteinemia (also called dysbetalipo-proteinemia). Diagnosis of this highly atherogenic type of dyslipoproteinema fails with measurement of TC, TG, HDL-C and LDL-C, and should include measurement of apoB [10].
Apo(a)
Apo(a), an apo specific for Lp(a), is highly polymorphic in size due to a variable number of kringle IV type 2 (KIV-2)-encoding sequences, producing >40 apo(a) isoforms varying in number among individuals and populations. Lp(a) is a highly prevalent, genetic risk-enhancing factor for CVD and calcified aortic valve disease [11], which is proatherogenic, proinflammatory and prothrombotic. Statin therapy does not lower Lp(a)-cholesterol. A novel Lp(a)-lowering therapy using an antisense oligonucleotide directed against mRNA of apo(a) induced a decrease of Lp(a) by approximately 70.% [12].
ApoAI
ApoAI is the major apo in HDL and involved in many of the anti-atherogenic functions of HDL. One of them is the reverse cholesterol transport by HDL, whereby excess cholesterol in peripheral tissues is carried back to the liver for excretion. Individuals with low apoAI (<1.2g/L) have higher risk of CVD than those with high apoAI concen-tration (>1.6.g/L). However, genetic studies have shown that the latter correlation may not be due to the apoAI elevation itself, but may be caused by the concomitant reduc-
tion in TG concen-trations and thus by the concomitant reduction in atherogenic remnants lipoproteins [13].
Apo profiles, cardiovascular risk and cardiovascular precision care
There is now ample evidence that apo pro-filing may improve CVD risk stratification. ApoB is superior to LDL-C and non-HDL-C (which equals TC minus HDL-C) as a predictor of CVD risk [14], indicating that the number of atherogenic apoB100-containing particles relates more to risk of CVD than the mass of cholesterol in these particles.
Others have used apoB/apoAI as this ratio is accurate in risk prediction, particularly among high-risk individuals. We reported that the apoB/apoAI ratio was strongly associated with risk of myocardial infarction [15]. In patients with excess remnants, high concentrations of apoCI, apoCII, apoCIII and apoE can be found in those particles. ApoCII, apoCIII, and apoE were sig-nificantly associated with incident CVD in the Bruneck Study [16]. These associations remained significant after adjustment for traditional risk factors, and demonstrate the advantage of using apos to identify dyslipidemic states, particularly those related to excess remnants.
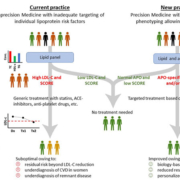
While the current clinical care pathway does indeed improve CVD risk, it does not allow for personalized treatment. Current generic treatments often encompass statins which predominantly address LDL-C reduction. Yet, considerable residual risk remains, particularly in women, in individuals with small dense LDL [2], in individuals with excess remnant particles [10], and in those with elevated Lp(a) [11]. Based on our current understanding of lipoprotein patho-physiology and the knowledge acquired from Mendelian randomization trials and protein-based outcome studies, we suggest that a multiplexed panel including apoAI, apoB, apoCI, apoCII, apoCIII, apoE and apo(a) could provide improved risk stratification (Fig. 1). We argue that implementation of a biology-driven multiplexed apo panel will provide better CVD risk stratification and allow for personalized treatment, in which patients are treated with effective biology-based treatments [17]. This is feasible from a technological perspective, as the mass-spectrometry (MS)-based test we developed [5] revealed stable analytical performance over a long period of time [18] which avoids problems with (a) poor analytical selectivity of direct LDL-C and HDL-C methods, (b) considerable heterogeneity of lipoprotein particle composition and the measurands of interest, and (c) the use of calculated LDL-C methods [19]. The apo profiles will provide insight in the pathophysiology of the dysli-pidaemia. Awaiting the evidence from clinical trials, it can be anticipated that the multiplexed serum apo panel will facilitate the refining of the diagnosis and will allow the use of targeted personalized therapies such as monoclonal antibodies and antisense oligo-nucleotides. We conclude that the serum apo panel has the potential to address unmet clinical needs, i.e. residual CV risk, and to catalyse precision cardiovascular diagnostics and therapy in familial dyslipidaemia patients at high risk.
References
1. Monaghan PJ, Lord SJ, St John A, Sandberg S, Cobbaert CM, Lennartz L, Verhagen-Kamerbeek WD, Ebert C, Bossuyt PM, et al. Biomarker development targeting unmet clinical needs. Clin Chim Acta 2016; 460: 211–219.
2. Langlois MR, Chapman MJ, Cobbaert C, Mora S, Remaley AT, Ros E, Watts GF, Borén J, Baum H, et al. Quantifying atherogenic lipoproteins: current and future challenges in the era of personalized medicine and very low concentrations of LDL cholesterol. A consensus statement from EAS and EFLM. Clin Chem 2018; 64: 1006–1033.
3. Hood L. Systems biology and P4 medicine: past, presence and future. Rambam Maimonides Med J 2013; 4; e0012.
4. Sniderman A, Couture P, de Graaf J. Diagnosis and treatment of apolipoprotein B dyslipoproteinemias. Nat Rev Endocrinol 2010; 6: 335–346.
5. Van den Broek I, Romijn FP, Nouta J, van der Laarse, A, Drijfhout JW, Smit NP, van der Burgt YE, Cobbaert CM, et al. Automated multiplex LC-MS/MS assay for quantifying serum apolipoproteins A-I, B, C-I, CII, C-III, and E with qualitative apolipoprotein E phenotyping. Clin Chem 2016; 62: 188–197.
6. Varbo A, Benn M, Tybjærg-Hansen A, Jørgensen AB, Frikke-Schmidt R, Nordestgaard BG. Remnant cholesterol as a causal risk factor for ischemic heart disease. J Am Coll Cardiol 2013; 61: 427–436.
7. Graham MJ, Lee RG, Bell TA, 3rd, Fu W, Mullick AE, Alexander VJ, Singleton W, Viney N, Geary R, et al. Antisense oligonucleotide inhibition of apolipoprotein C-III reduces plasma triglycerides in rodents, nonhuman primates, and humans. Circ Res 2013; 112: 1479–1490.
8. Pollin TI, Damcott CM, Shen H, Ott SH, Shelton J, Horenstein RB, Post W, McLenithan JC, Bielak LF, et al. A null mutation in human APOC3 confers a favorable plasma lipid profile and apparent cardioprotection. Science 2008; 322: 1702–1705.
9. Jørgensen AB, Frikke-Schmidt R, Nordestgaard BG, Tybjærg-Hansen A. Loss-of-function mutations in APOC3 and risk of ischemic vascular disease. N Engl J Med 2014; 371: 32–41.
10. Sniderman AD. Editorial. Type III hyperlipoproteinemia: the forgotten, disregarded, neglected, overlooked, ignored but highly atherogenic, and highly treatable dyslipoproteinemia. Clin Chem 2019; 65: 225–227.
11. Nordestgaard BG, Langsted A. Lipoprotein (a) as a cause of cardiovascular disease: insights from epidemiology, genetics, and biology. J Lipid Res 2016; 57: 1953–1975.
12. Viney NJ, van Capelleveen JC, Geary RS, Xia S, Tami JA, Yu RZ, Marcovina SM, Hughes SG, Graham MJ, et al. Antisense oligonucleotides targeting apolipoprotein(a) in people with raised lipoprotein(a): two randomised, double-blind, placebo-controlled, dose-ranging trials. Lancet 2016; 388: 2239–2253.
13. Haase CL, Tybjaerg-Hansen A, Grande P, Frikke-Schmidt R. Genetically elevated apolipoprotein A-I, high-density lipoprotein cholesterol levels, and risk of ischemic heart disease. J Clin Endocrinol Metab 2010; 95: E500–E510.
14. Walldius G, Jungner I, Holme I, Aastveit AH, Kolar W, Steiner E. High apolipoprotein B, low apolipoprotein A-I, and improvement in the prediction of fatal myocardial infarction (AMORIS study): a prospective study. Lancet 2001; 358: 2026–2033.
15. Bodde MC, Hermans MPJ, Jukema JW, Schalij MJ, Lijfering WM, Rosendaal FR, Romijn FPHTM, Ruhaak LR, van der Laarse A, Cobbaert CM. Apolipoproteins A1, B, and apoB/apoA1 ratio are associated with first ST-segment elevation myocardial infarction but not with recurrent events during long-term follow-up. Clin Res Cardiol 2019; 108: 520–538.
16. Pechlaner R, Tsimikas S, Yin X, Willeit P, Baig F, Santer P, Oberhollenzer F, Egger G, Witztum JL, et al. Very-low-density lipoprotein associated apolipoproteins predict cardiovascular events and are lowered by inhibition of APOC-III. J Am Coll Cardiol 2017; 69: 789–800.
17. Ruhaak LR, van der Laarse A, Cobbaert CM. Apolipoprotein profiling as a personalized approach to the diagnosis and treatment of dyslipidemia. Ann Clin Biochem 2019; 56: 338–356.
18. Ruhaak LR, Smit NPM, Romijn F, Pieterse MM, van der Laarse A, van der Burgt YEM, Cobbaert CM. Robust and accurate 2-year performance of a quantitative mass spectrometry-based apolipoprotein test in a clinical chemistry laboratory. Clin Chem 2018; 64: 747–749.
19. Langlois MR, Descamps OS, van der Laarse A, Weykamp C, Baum H, Pulkki K, von Eckardstein A, De Bacquer D, Borén J, et al; EAS-EFLM Collaborative Project. Clinical impact of direct HDLc and LDLc method bias in hypertriglyceridemia. A simulation study of the EAS-EFLM Collaborative Project Group. Atherosclerosis 2014; 233: 83–90.
20. Conroy RM, Pyörälä K, Fitzgerald AP, Sans S, Menotti A, De Backer G, De Bacquer D, Ducimetière P, Jousilahti P, et al. Estimation of ten-year risk of fatal cardiovascular disease in Europe: the SCORE project. Eur Heart J 2003; 24: 987–1003.
The authors
L. Renee Ruhaak1 PhD, Arnoud van der Laarse1,2 PhD, Christa M. Cobbaert*1 PhD
1 Department of Clinical Chemistry and Laboratory Medicine, Leiden University Medical Center, Leiden, The Netherlands
2 Department of Cardiology, Leiden University Medical Center, Leiden, The Netherlands
*Corresponding author
E-mail: c.m.cobbaert@lumc.nl
Figure 1. Proposed shift to precision medicine and personalized treatment.
Conceptual view of the proposed paradigm shift from traditional population-based medicine (left) to precision medicine with personalized therapies (right) employing clinical test-treatment pathways for risk reduction according to current (left) and new (right) practices (based on the clinical pathway technique outlined by Monaghan et al. [1]).
Dx, diagnosis; Tx, therapy; TTx, targeted therapy; SCORE, Systemic Coronary Risk Estimation, a widely applied risk assessment system [20]. Adapted from Ruhaak LR, van der Laarse A, Cobbaert CM. Apolipoprotein profiling as a personalized approach to the diagnosis and treatment of dyslipidemia. Ann Clin Biochem 2019; 56: 338–356 [17].