Monitoring therapeutic drug dosage in rheumatic diseases, chronic inflammatory bowel diseases and psoriasis
by Dr Jacqueline Gosink
Tumour necrosis factor (TNF) inhibitors are increasingly used to treat chronic inflammatory bowel diseases, rheumatic diseases and psoriasis. Continuous drug level monitoring and administration interval adjustment accompanying the treatment with TNF inhibitors is the key to successful, individualized and targeted patient care. Concentrations of the TNF inhibitors adalimumab or infliximab can be specifically measured by serological assays. One possible cause of a decrease in drug efficacy is the generation of antibodies against adalimumab or infliximab and sensitive detection of these anti-drug antibodies is also possible. Monitoring drug levels may contribute to effective and successful therapeutic treatment strategy.
Introduction
In autoimmune diseases, such as chronic inflammatory bowel diseases (Crohn’s disease, ulcerative colitis), rheumatic diseases (rheumatoid arthritis, spondyloarthrosis) and psoriasis, the individual’s immune response is directed against its own healthy cells and tissues. During the misdirected inflammatory response, macrophages produce and release the pro-inflammatory cytokines such as TNF, which induces fever, apoptotic cell death and inflammation. Treatment with TNF inhibitors interrupts the inflammatory response by neutralization of TNF activity. TNF inhibitors are biologics in form of monoclonal antibodies that bind mono specifically to TNF. Prominent representatives of monoclonal antibodies against TNF are infliximab (IFX) and adalimumab (ADL). IFX is a chimeric monoclonal antibody and acts as the active ingredient of Remicade®, which was the first drug from the group of TNF inhibitors to be approved for therapeutic use in 1998. ADL is an entirely human monoclonal antibody and is the active ingredient of Humira®. It was first approved in 2002 and can be administered subcutaneously by the patients themselves. As an economic alternative to the original prepar-ations, biosimilars are available for both drugs.
Therapeutic drug monitoring
Autoimmune diseases such as rheumatic diseases, chronic inflammatory bowel diseases and psoriasis cannot be cured. Instead, patients are treated symptomatically with antiinflammatory drugs, such as TNF inhibitors. However, the clinical reality shows that patients exhibit deviant responses to these biologics, as pharmacokinetics and pharmacodynamics differ from patient to patient and fluctuations occur in time. Pharmacokinetics includes the absorption, distribution, metabolism and excretion of the drug, whereas pharmacodynamics describes biochemical and physiological mechanisms of action and effects of a drug within the human body.
Therapeutic drug monitoring (TDM) can be used to manage and optimize drug administration based on measurement of drug concentration [1]. TDM is beneficial if dosage of a drug shows high pharmacokinetic variability and has an optimal therapeutic dose which may show clear reduction of symptoms with some delay after start of treatment.
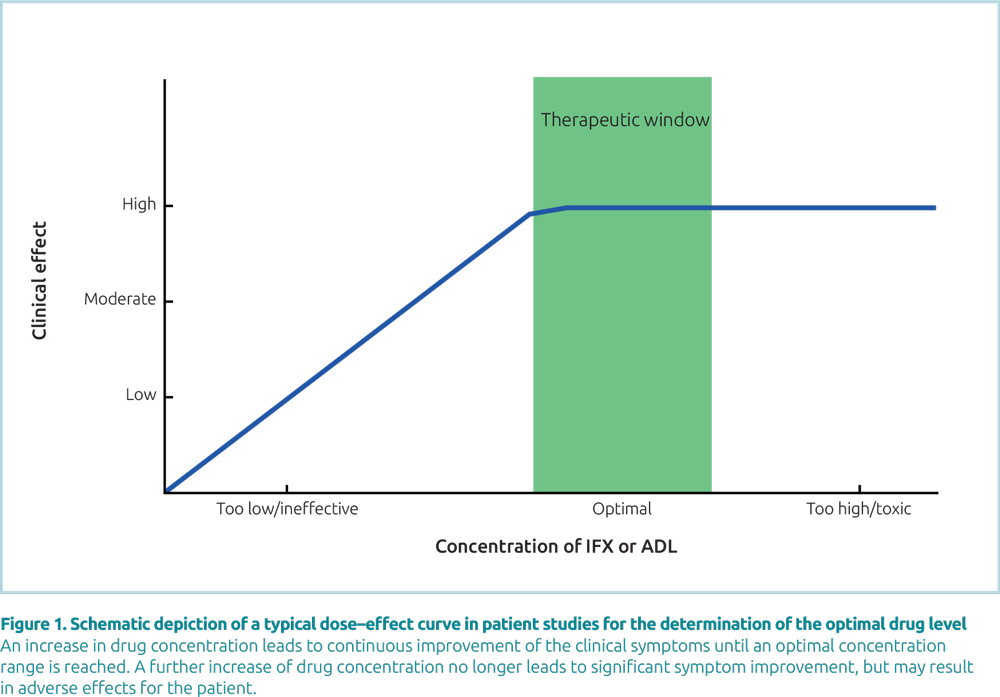
In ADL or IFX treatment, an increase in the drug level generally leads to continuous improvement of the clinical symptoms until an optimal concentration range is reached (plateau effect). In this plateau, further increase of the drug level no longer leads to symptom improvement (Fig. 1). To reach the optimal range, the drug dosage must be adjusted individually by measuring the effective drug concentration in blood. In several studies, test systems from Sanquin have been used for successful monitoring of optimal ADL and IFX concentrations [2–5]. For precise monitoring of the drug level, TDM should be initiated at an early stage and kept up for successful treatment and also continued after adjustment of the drug dose to the optimal range. Continuous drug level monitoring accompanying treatment with TNF inhibitors is key to individualized, targeted and cost-efficient patient care [6, 7].
Strategies of TDM: Proactive and reactive TDM
In proactive TDM, drug levels are measured in a scheduled, prophylactic manner aiming to optimize therapy to avoid supratherapeutic drug levels and prevent future disease activity. Proactive TDM is explicitly performed also in patients who show either a significant improvement in symptoms or persisting symptoms without any significant deterioration after therapy initiation. In the event of supratherapeutic drug levels, these can be recognized promptly and corrected by reducing the dosage [8] and/or administration intervals to avoid preventable side effects and successfully reduce the strain on the patient. MabTrack Level ELISAs (Sanquin Reagents, distributed worldwide by EUROIMMUN) can be used to precisely determine the concentration of IFX or ADL in patient serum or plasma (Fig. 2).
Treatment of patients in whom an optimal concentration of the active agent can be observed may be continued with the same or even a reduced drug dosage. A retrospective study provided evidence that proactive TDM enables identification of patients responding to the therapy (responders) and those who won’t respond (non-responders), even at an early therapy stage [9]. In contrast, reactive TDM is exclusively applied in non-responders to adjust the treatment as required. It allows investigation of possible causes for subtherapeutic levels, i.e. insufficient therapy adherence or the necessity of higher doses and/or interval reduction due to the patient’s pharmacokinetics/ pharmacodynamics.
Anti-drug antibodies
Another cause for insufficient clinical effectiveness may be the presence of specific antibodies against the drug (anti-drug antibodies, ADA). Despite the good overall effectiveness of IFX or ADL, a substantial number of patients form ADA [10], which can hamper the effect of the drug when reaching high levels.
ADA may circulate in the blood as either the free form or bound to the respective drug. Low levels of ADA form few immune complexes with the drug which is primarily still unbound and active [11]. With increasing ADA formation, the amount of immune complexes also rises. Eventually, at high ADA concentrations, all drug is bound in immune complexes and additional free ADA are present. At this point, active drug levels drop and treatment efficacy is dramatically impaired. However, studies indicate, that the detection of ADA does not fully correlate to the clinical symptoms in the tested patients [12, 13].
MabTrack Anti-Drug Antibody ELISAs (Sanquin Reagents BV), which enable specific detection of antibodies against infliximab or adalimumab as well as against their biosimilars, are easy to perform and can be fully automated, exclusively detect free (and usually more clinically relevant) ADA. Therefore, following the TDM strategy concerted with the Sanquin MabTrack Level ELISAs, ADA levels shall only be determined in case of drug levels dropping below a threshold of 0.5 μg/ml.
Summary
Thus, in addition to assessment of clinical symptoms, a combination of both regular drug monitoring and determination of ADA supports decisions about adjustments of the drug dosage and an early switch to other therapeutics in non-responders in order to improve the therapeutic effectiveness.
The author
Jacqueline Gosink PhD
EUROIMMUN AG, Seekamp 31, 23560 Lubeck, Germany
References
- Hallworth M. Chapter 39 Therapeutic drug monitoring. In: Marshall WJ, Lapsley M, Day AP, Ayling R, editors. Clinical Biochemistry: Metabolic and clinical aspects, pp. 767–786 (third edition). Churchill Livingstone 2014.
- Pouw MF, Krieckaert CL, Nurmohamed MT, et al. Key findings towards optimising adalimumab treatment: The concentration-effect curve. Ann Rheum Dis 2015; 74(3): 513–518.
- Menting SP, Coussens E, Pouw MF, et al. Developing a therapeutic range of adalimumab serum concentrations in management of psoriasis a step toward personalized treatment. JAMA Dermatol 2015; 151(6): 616–622.
- Vande Casteele N, Ferrante M, Van Assche G, et al. Trough concentrations of infliximab guide dosing for patients with inflammatory bowel disease. Gastroenterology 2015; 148(7): 1320–1329.
- van den Bemt BJF, den Broeder AA, Snijders GF, et al. Sustained effect after lowering high-dose infliximab in patients with rheumatoid arthritis: a prospective dose titration study. Ann Rheum Dis 2008; 67(12): 1697–1701.
- Yao J, Jiang X, You JHS. A systematic review on cost-effectiveness analyses of therapeutic drug monitoring for patients with inflammatory bowel disease: from immunosuppressive to anti-TNF therapy. Inflamm Bowel Dis 2020; doi: 10.1093/ibd/izaa073. Epub ahead of print.
- Krieckaert CLM, Nair SC, Nurmohamed MT, et al. Personalised treatment using serum drug levels of adalimumab inp atients with rheumatoid arthritis: an evaluation of costs and effects. Ann Rheum Dis 2015; 74(2): 361–368.
- l’Ami MJ, Krieckaert CL, Nurmohamed MT, et al. Successful reduction of overexposure in patients with rheumatoid arthritis with high serum adalimumab concentrations: an open-label, non-inferiority, randomised clinical trial. Ann Rheum Dis 2018; 77(4): 484–487.
- Jurado T, Plasencia-Rodríguez C, Martínez-Feito A, et al. Predictive value of serum infliximab levels at induction phase in rheumatoid arthritis patients. Open Rheumatol J 2017; 11: 75–87.
- Mok CC, van der Kleij D, Wolbink GJ. Drug levels, anti-drug antibodies, and clinical efficacy of the anti-TNFα biologics in rheumatic diseases. Clin Rheumatol 2013; 32(10): 1429–1435.
- van Schouwenburg PA, Rispens T, Wolbink GJ. Immunogenicity of anti-TNF biologic therapies for rheumatoid arthritis. Nat Rev Rheumatol 2013; 9(3): 164–172.
- Van Stappen T, Vande Casteele N, Van Assche G, et al. Clinical relevance of detecting anti-infliximab antibodies with a drug-tolerant assay: post hoc analysis of the TAXIT trial. Gut 2018; 67(5): 818–826.
- Ruwaard J, Marsman AF, Nurmohamed MT, et al. Antidrug antibody detection for adalimumab depends on the type of assay used: an experimental approach to help clinicians interpret diagnostic data. Clin Exp Rheumatol 2019; 37(5): 756–761.