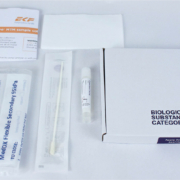
EKF launches PrimeStore MTM pathogenic sample collection and transportation kit
EKF Diagnostics, the global in vitro diagnostics company, has launched a PrimeStore MTM (molecular transport media) sample collection kit. This new kit enables convenient and safe collection, transportation and handling of pathogenic samples, including COVID-19 and influenza. Two kit configurations are now available: a postal sample kit ideal for home use and a multi-pack of sample kits suitable for on-site mass sampling. To meet growing demand for this sample collection device, EKF has increased its manufacturing capacity in the UK and mainland Europe.
Pathogenic samples stored in the FDA-cleared, CE-IVD marked PrimeStore MTM novel viral transport media, including SARS-CoV-2, are inactivated within minutes of collection. This allows such samples to be transported safely through the postal system or via courier and complies with UN3373 packaging regulations.
Furthermore, as samples are stable in PrimeStore MTM for seven days at ambient temperatures, this removes the need for all cold chain logistics during sample collection, transportation and storage. Thereby, preventing mishandled samples and making postal collection a realistic option.
The new sample collection kit pairs PrimeStore molecular transport media in a cryovial with a sterile flocked swab (for nasal and oral sampling) in a CE marked kit, which also includes a UN3373 95kPa compliant specimen collection bag, absorbent pad and instructions for use. In addition, the postal sample collection kit includes a UN3373 compliant postal box with security seal for sample return and postal pouch for collection kit delivery.
For more information, visit: www.ekfdiagnostics.com
Supplier: EKF Diagnostics
Website: https://www.ekfdiagnostics.com/