New triterpene discovery overturns current knowledge of highly useful compound, opening up new world for drug discovery
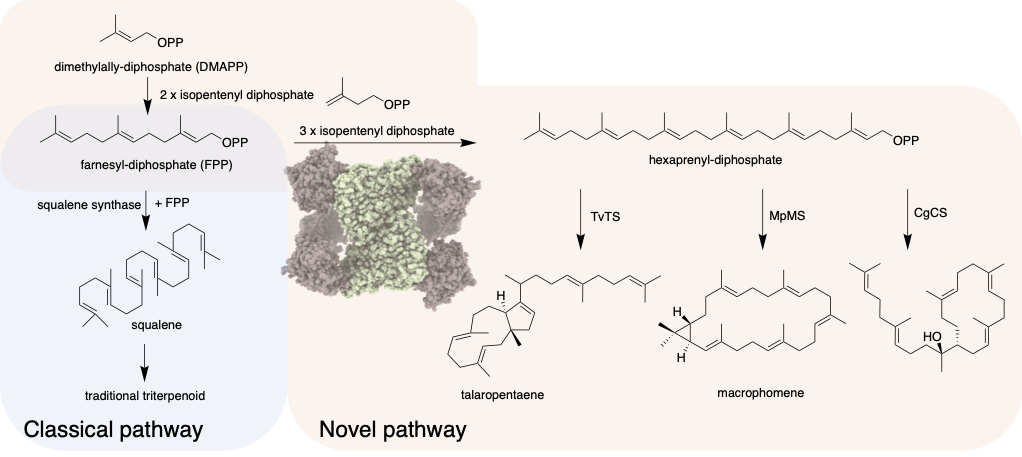
A remarkable discovery and collaborative effort have revealed a new type of triterpenes, a group of organic compounds which are an important source of many medicines. Until now, all triterpenes were believed to be derived from squalene, itself a type of triterpene. However, for the first time, researchers witnessed biosynthesis, the formation of complex compounds from simple ones in living organisms, of triterpenes in fungi without the use of squalene. This important discovery opens up a whole new world of possibilities for pharmaceutical science.
Triterpenes are organic compounds which are found abundantly in animals, plants, micro-organisms and even us. About 20,000 different triterpenes have been found and they are widely used in cosmetics, food supplements and, most importantly, medicine, thanks to their anti-inflammatory, anti-cancer, anti-diabetic and other valuable properties. Until now, all known triterpenes were thought to be generated from a common precursor, squalene.
However, as revealed in Nature, a collaborative effort among the University of Tokyo and KEK in Japan, Wuhan University in China and Bonn University in Germany, has found a new type of triterpenes that doesn’t require squalene.
“Nobody could have imagined this happening in nature. This is the discovery of a new biosynthetic machine,” explained Professor Ikuro Abe from the Graduate School of Pharmaceutical Sciences at the University of Tokyo.
Often, multiple enzyme reactions are needed to create complex molecular compounds, such as when our bodies use squalene to create hormones and bile acid. However, in just a single enzyme reaction, a simple molecule called a C5 isoprene unit, or building block, was the starter to construct a very complex triterpene molecular structure.
The discovery happened almost by accident by the team at Wuhan University, who were working on genome mining to find new natural products. They were not necessarily searching for triterpenes, but they found new genes widely distributed in fungi. “They didn’t know the genes’ function,” said Abe. “So they did a characterization of these news genes, and one of them happened to be triterpene synthesis.”
That is when they asked the other teams to get involved. According to Abe, the team at Bonn University are good at chemistry, so they worked on elucidating the detailed enzyme reaction mechanism, and researchers at the University of Tokyo and KEK applied their expertise in structural analysis. He said that once you understand the structure, you can modify it. “Change something here or there and see what happens. We can understand the structure-function relationship. It’s like a puzzle,” he explained.
“Chemistry in nature is more efficient than the chemical synthesis we use in industry. That is why we are interested in the biosynthetic processes done in nature,” Abe said. “Nature’s method is a better, cheaper and cleaner process. We are trying to better understand how processes take place in nature so that we can recreate or redesign it in the lab, to get more and more important and useful compounds.”
This new discovery is only the beginning. “Now that we have solved the protein structure, we are already manipulating the biosynthetic machinery to try to produce more useful molecules, for example, for drug development,” Abe said.
Reference
Hui Tao, Lukas Lauterbach, Guangkai Bian, et. al. “Discovery of non-squalene triterpenes” Nature, 1 June 2022.doi: https://doi.org/10.1038/s41586-022-04773-3