Patient home test kit for calprotectin
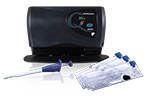
IBDoc is the first in-vitro diagnostic test to measure the fecal inflammation marker calprotectin at home. IBDoc just received a CE mark for patient self testing and is a revolutionary new test for gut inflammation aimed at people with inflammatory bowel disease, Crohn’s and ulcerative colitis. IBDoc eliminates the need for patients to bring stool samples to the clinic for analysis. Instead, its simple to use technology combining a stool extraction device CALEX, a calprotectin test strip and the IBDocsmart phone app to read the test result by state-of-the-art image processing, allows IBD patients to actively participate in their disease management and communicate with the healthcare providers via the internet as well. The backbone of this home test system is the IBDoc web portal for patient data management that keeps the IBD patient under constant vigilance of the healthcare professional for real time support. The IBDoc portal is designed with the highest security standards to keep confidential patient data safe. Calprotectin measured in stool is a non-invasive biomarker for gastrointestinal inflammation. It correlates well with lesions and with endoscopic scores of disease activity in both, Crohn’s disease and ulcerative colitis. Fecal calprotectin is superior to classically used blood biomarkers, such as C-reactive protein (CRP), erythrocyte sedimentation and others.