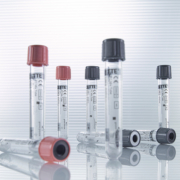
Vacuum sample tube with glycolysis inhibition
The rapid breakdown of glucose (glycolysis) in venous blood samples is very significant for the diagnosis of both diabetes mellitus and gestational diabetes which should be detected at an early stage to avoid complications such as infections, premature births and long-term effects for the mother and child. In order to have a reliable diagnosis, it is necessary to inhibit glucose breakdown immediately after collecting blood. Various institutions have drafted guidelines, which recommend the addition of a citrate-fluoride additive to maintain the “in vivo glucose level”. The special feature of the new VACUETTE FC Mix tube from Greiner Bio-One is the powder additive. It stabilizes the glucose level immediately after collection for 48 hours. This allows for reliable diagnosis of diabetes conditions and avoids false negatives. The stabilization is carried out in the whole blood and therefore does not require immediate centrifugation. Unlike in tubes where liquid is added, the finely granulated additive does not cause a dilution effect. There is no need to convert the measurement result. The citrate/citric acid buffer reduces the pH value in the sample. As a result, the enzymes needed for the glycolysis process are inhibited and the actual “in vivo level” is stabilized from the start. The additive is completely dissolved, and therefore optimally mixed with the sample, after swivelling ten times. In the case of storage between 4°C and room temperature, a further sodium fluoride additive ensures long-term stabilization for 48 hours. The VACUETTE FC Mix tube is available with both a grey and pink security cap and therefore allows for differentiation from standard glucose tubes. The cap is particularly easy to open and allows for hygienic working in the laboratory. The VACUETTE FC Mix tube is made of highly-transparent PET plastic and is shatter-proof.
Read more