Carbapenemase variants detection test
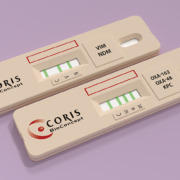
Surveillance and control of resistance to beta-lactam among gram-negative bacteria remains nowadays a big concern for microbiologists worldwide. Descriptions of OXA-48 associated infections are still revealing huge impact on antimicrobial treatment and patient morbidity. For clinical laboratories, OXA-48 and its variants represent the most challenging resistance mechanism to detect and are often under-detected by routine diagnostic methods. Multiple allelic variants of OXA-48 have been described, with diverse hydrolytic profiles. As an example, the single amino acid substitution and four amino acid deletion described in OXA-163 confer an increased hydrolytic activity against cephalosporins, coupled to a loss of resistance to carbapenems. This particular hydrolytic profile makes detection confusing. Treatment failures can impede patient recovery and favour the selection of new resistant strains. Until recently, efficient identification of OXA-163-expressing strains relied on gene sequencing, which is not practical for a clinical laboratory daily routine. The recently available immunochromatographic lateral flow assay developed by Coris BioConcept to specifically differentiate OXA-163 from OXA-48 proteins, replaces costly and lengthy genotypic methods. This phenotypic test is an easy-to-use and instrument-free test that provides a fast (15 minutes) and accurate (100%) identification of variants. Precise detection of other carbapenemases will be included in the test to reach a 5-parameters assay. This up-coming “RESIST-5” test commercialized by the same company is the ideal tool to precisely identify the most prevalent carbapenemase-mediated resistance mechanisms on a single phenotypical assay. Its use on a daily routine in healthcare facilities will enable a broader surveillance of worldwide-disseminating new carbapenemase variants and help clinicians in the daily patient care.