ELITechGroup’s Staining Technology Expands with Aerospray® Cytology Slide Stainer/Cytocentrifuge
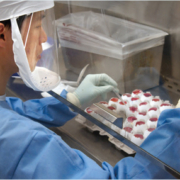
Logan, UT, January 17, 2020 – ELITechGroup Inc., recognized worldwide as slide staining and cytocentrifuge technologic pioneers and innovators, with nearly 30 patents, over the 35-year lifespan of Aerospray staining technology, announces the launch of the Aerospray Cytology Slide Stainer Cytocentrifuge. The ELITechGroup’s Aerospray Cytology Slide Stainer/Cytocentrifuge automates the preparation and staining of liquid based cytology specimens to facilitate the examination of an array of cell differentiations utilizing Papanicolaou staining techniques. The advantages of the Aerospray Cytology Slide Stainer/Cytocentrifuge is that the device performs two functions of sample preparation through cytocentrifugation and sample staining. The system maximizes cell recovery to the slide through the use of its Cytopro® Rotor, eliminates cross-contamination with an application of fresh stain for each slide, and provides programmable regressive and progressive staining capabilities that can be stored in memory to meet the needs and preferences of any end user. The instrument standardizes staining across multiple labs, provides consistently high-quality control, is low maintenance, reduces the labor/cost per slide, enables laboratory compliance, and is the perfect back-up for high automation laboratories for specialty staining and during maintenance. “ELITechGroup’s portfolio of Aerospray slide stainers and cytocentrifuges has enabled the company to develop a worldwide reputation for performance and reliability. Our new Aerospray Cytology Stainer/Cytocentrifuge proves our commitment to ongoing innovation by expanding into new laboratories where we can offer superior staining results,” says Bryce McEuen, Vice President, Biomedical Systems. ELITechGroup will be showcasing the Aerospray Cytology Slide Stainer/Cytocentrifuge at key industry events throughout the year of 2020. Learn more at AerosprayStaining.com.
About ELITechGroup
ELITechGroup is a privately held group of worldwide manufacturers and distributors of in vitro diagnostic equipment and reagents for clinical systems, microbiology, molecular diagnostics, hematology, cytology, and sweat test systems. Their mission is to improve patient care by empowering laboratories to do more in less time, with accuracy, to enable rapid and accurate course of treatment for patients.
Read more