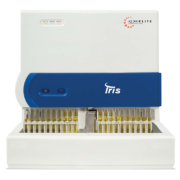
The Iris iRICELL workcell integrates urine chemistry and microscopy into a fully automated walk-away urinalysis system that is easy to use and maintain. The integrated workcell combines the iQ200 Series automated microscopy system with the iChemVELOCITY automated urine chemistry system. The iQ200 Series delivers clear, clinically relevant urine particle images that are auto-classified for more objective and consistent results. This automated microscopy system is designed for all volume workloads, delivering a shorter turnaround time and standardizing results. The iQ200 Series are available either as a stand-alone system or connected to an iChemVELOCITY urine chemistry analyser to form an automated iRICELL workcell. The iQ200SPRINT is one of the fastest automated systems on the market, meeting high-volume productivity and workload requirements. It can handle 101 microscopic samples an hour. The IQ200ELITE manages medium- to high-volume workloads running 70 microscopic samples an hour; with the iQ200SELECT more suitable for lowvolume workloads running 40 microscopic samples an hour. iChemVELOCITY provides a fully automated, high-capacity urine chemistry analysis with excellent low-end sensitivity. The system delivers a high throughput of 210 samples an hour, with the continuous strip loading and a capacity of 300 strip loads. It has a pad on the strip designed to detect and measure the presence of ascorbic acid. This provides clinically significant information about potential interference with key chemistry assays. The system evaluates all standard urine chemistry parameters, including glucose, protein, bilirubin, urobilinogen, pH, specific gravity, blood, ketones, nitrite and leukocyte esterase.
Utility of reflex urine culture based on results of urinalysis and automated microscopy
Specimens submitted for urine culture in hospital settings are frequently negative for bacteria. Various approaches have been developed to select urine samples in the laboratory to improve the efficiency of handling these samples. At a time of concern for cost containment, utilization of a reflex testing policy using specific screening criteria would be beneficial to eliminate unnecessary urine cultures. With this in mind, an evaluation of Beckman Coulter’s IRIS iQ200 system (urinalysis and automated urine microscopy) was carried out in the US at the Johns Hopkins Bayview Medical Center (JHBMC) clinical microbiology laboratories. The study prospectively collected and reviewed 1248 clinical urine specimens submitted for urinalysis and/or urine culture. It investigated the IRIS iQ200’s utility in aiding a predictive algorithm for the implementation of reflex urine cultures. Findings showed that these test parameters, separate or combined, may be a useful screening method to determine the need for a reflex urine culture.
Read more