Human NGAL monomer-specific ELISA Kit
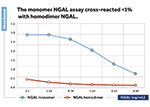
BioPorto’s new ELISA Kit will enable researchers to measure the monomeric form of NGAL with a highly specific assay for monomeric NGAL, which cross-reacts less than 1 % with the homo-dimer form of NGAL. During the past 5 years, researchers around the world have discussed and debated different theories about NGAL molecular forms (monomer, homodimer, heterodimer). The discussions have been about which of the forms are the predominant in different clinical situations, and also if one form is more specific for one clinical diagnosis than the other. BioPorto is now making the unique monomer-specific ELISA kit publicly available. This is the first monomer-specific NGAL assay on the market. The assay has already been used in two communications, at poster level, at the annual meeting of the American Society of Nephrology in both 2012 and 2013 (Bagert et al). The assay is for research use only, not for use in diagnostic procedures.
Read more