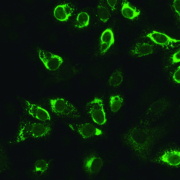
ELISA and indirect immunofluorescence tests (IIFT) have been developed for sensitive and specific detection of antibodies against Zika virus in patient serum samples. The assays are suitable for diagnosing acute infections as well as for disease surveillance. In particular, serological analyses can aid the differentiation of infections with Zika virus, dengue virus and chikungunya virus, which manifest with similar symptoms and are endemic in much the same geographic regions. Anti-Zika Virus ELISA (IgM or IgG) are based on highly specific recombinant Zika NS1 protein which avoids cross reactivity with other flaviviruses. Data from panels of well characterized sera have confirmed that there is no cross reactivity with flaviviruses including dengue, West Nile, yellow fever and Japanese encephalitis viruses. In studies on clinically and serologically characterized samples the IgM and IgG ELISA showed 100% sensitivity and 100% specificity. Anti-Zika Virus IIFT (IgM or IgG) utilize Zika virus-infected cells as the antigenic substrate. Positive and negative results are evaluated by fluorescence microscopy. With the Arboviral Fever Mosaic 2 the Zika virus substrate is incubated in parallel with substrates for chikungunya virus and dengue virus serotypes 1 to 4. This BIOCHIP combination can help in the differential diagnosis of Zika, dengue and chikungunya virus infections. Due to the use of whole virus particles, cross reactivities between flavivirus antibodies can occur. Serological tests provide a longer window for diagnosis than direct detection methods, which are only effective during the viremic phase within the first week after onset of symptoms. Detection of specific IgM or a significant rise in specific IgG in a pair of samples taken seven to ten days apart is evidence of an acute infection. Serological analyses are also important for prenatal monitoring, screening of donated blood and epidemiological studies. Zika virus is the pathogenic agent of Zika fever, an infectious topical disease which manifests with fever, exanthema and arthritis. Zika virus infection has been linked to congenital malformations, in particular microcephaly, and neurological complications such as Guillain-Barré syndrome. The virus is transmitted by mosquitoes of the Aedes family. Zika virus is currently spreading explosively in the Americas.
Read more