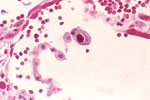
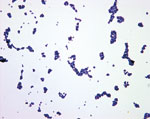
Five new infectious disease panels, including a first-of-its-kind performance panel for parvovirus B19 have been launched. The HIV Genotype Performance Panel provides researchers with a diverse set of samples, both geographically and in terms of genotypes: eight of the nine panel members are HIV positive, representing eight different HIV-1 genotypes from seven countries. Made from cultured virus diluted in defibrinated plasma, this unique panel is designed to help researchers, manufacturers and labs develop, evaluate and troubleshoot a variety of HIV assays, including those for HIV RNA and the newest screening tests for combined HIV antigen and antibody. The Worldwide HBV DNA Performance Panel is a 20-member panel including samples from 10 countries with HBV genotypes ranging from A to H. It delivers a broad range of data, with information on genotype, drug resistance mutations and test results for, HBV DNA, HBsAg and HbeAg. The Anti-Hepatitis A Virus Mixed Titer Performance Panel offers customers a 21-member panel that includes members positive for IgG and IgM, contains specimens collected from 2000 to 2010, and represents a diverse range of HAV reactivities (from low to medium to high). Data are available for both new and established test methods, providing greater applicability across a broad range of laboratory and research settings. The Anti-CMV (Cytomegalovirus) Mixed Titer Performance Panel provides 21 naturally occurring panel members with data from twelve commercially available anti-CMV assays. It also features a detailed graphical representation of test member results, making it easier than ever to find and utilise relevant data. These five new infectious disease panels, as well as a diverse selection of other infectious disease panels, are available immediately worldwide.
Read more